Graphical Abstract:

The first half of our year in STEM class has consisted of work on our "Independent Research Project." This project stays true to its name, and allows each and every Mass Academy student to select any problem or idea to research, design or create. My project relates to global warming, and specifically "Carbon Capture," which is any and all methods/technologies for removing CO2 from the environment. I aim to create a filtration device for removing CO2 from automobile exhaust emissions.
Overview:
Rising levels of CO2 in the atmosphere are driving climate change, with transportation emissions accounting for about 28% (EPA, 2023). The CO2 emissions of vehicles are increasing rapidly, as stated in a study by the European Parliament; “Transport is the only sector where greenhouse gas emissions have significantly increased in the past three decades, rising 33.5% between 1990 and 2019” (Parliament, 2019). Transportation is currently environmentally unsustainable, and there is little to no technology that aims to mitigate emissions from gas powered vehicles, especially at a commercially realistic price with availability to the average consumer.
Abstract:
Graphical Abstract:

Engineering Need:
There is currently little consumer-level technology that directly captures or removes CO₂ at the point of emission in cars.
Engineering Objective:
Design and prototype a chemical-scrubbing exhaust attachment that uses Hydroxide-based filtration to remove CO₂ from car exhaust streams.
Background:
Background Graphic:
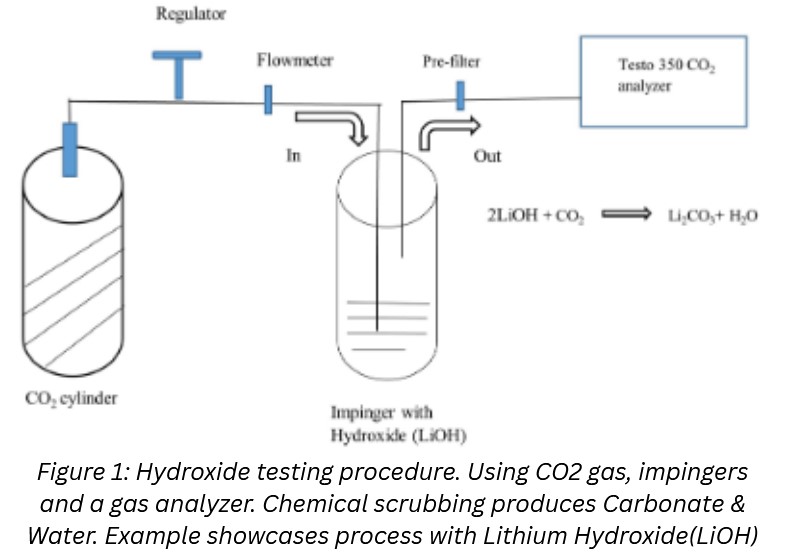
Methodology:
To complete the Hydroxide Testing, a cylinder filled with Nitrogen-gas mixture flowed into an impinger containing the desired hydroxide. Then the filtered gas flows into a gas analyzer to determine concentration of CO2 after filtration.
Using the same method, I then varied the CO2 concentration of the gas and tested filtration again at these higher concentrations using the hydroxide that was most efficient in the previous test.
Finally, I completed the same test with varied CO2 concentration again, but this time with two impingers to mimic a multi-layered filtration system.
Methodology Graphic:
Results:
Analysis:
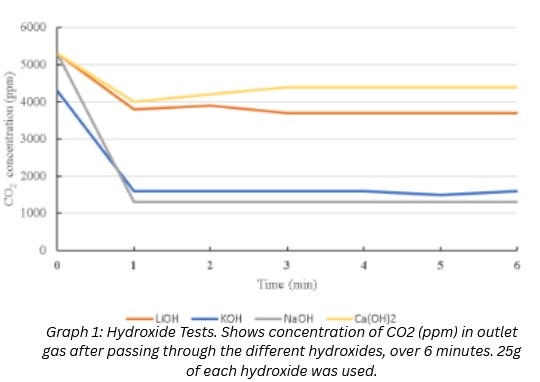
Test 1:
LiOH ~ 32.40% --- KOH ~ 68.60% --- NaOH ~ 74.5% --- Ca(OH)2 ~ 24.9%
This data showed that NaOH filtered most effectively at low CO2 concentrations. While this is a good sign, it does not accurately reflect how it will perform in a car exhaust application because car exhausts reach up to 15% CO2 concentration.
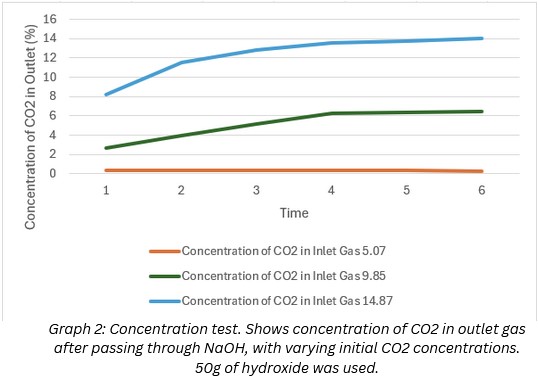
Test 2:
@5% ~ 93.2% --- @10% ~63.6% --- @15% ~ 25.1%
This data showed that NaOH is much less efficient at high concentrations. However, this was only tested with 1 layer of filtration, and a low amount of the hydroxide. Therefore the data reveals that more hydroxide and more filtration is needed for this method to function in a car exhaust application.
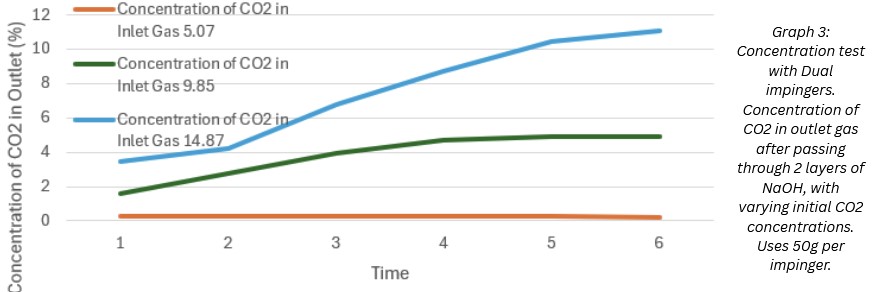
Test 3:
@5% ~ 95.6% --- @10% ~ 78.0% --- @15% ~ 51.2%
This data showed that multi-layered filtration greatly increases efficiency of NaOH's filtration. Thus proving my theory from the previous test, and showing signs of possible real-world use of this method of CO2 filtration on automobile exhausts.
Conclusion:
NaOH filtered CO2 most efficiently overall. This makes it the hydroxide that should be used in the real-world application.
Multi-layered hydroxide filtration improved CO2 removal. Higher amounts of hydroxide and more contact between the gas and the hydroxide allows for much better filtration, which should be utilized in the design of the real-world device.
Real world building and testing is still needed. While all the data suggests this project to be possible, the device is yet to be built or tested, meaning that the true success of this device is not yet guaranteed in a real-world application.
Future Steps:
First, I can further improve filtration capability with more layers and more hydroxide. There could be 3 thin compact mesh like filter trays, covered with 100g or more of the NaOH in powder form to maximize contact with the gas. This would improve filtration by a large amount and increase longevity.
Finally, I must actually construct and test the physical filter device in a real-world application. This would allow the true feasibility of this device to be revealed, and show important aspects such as longevity and durability.
References:
February (STEM) Poster: