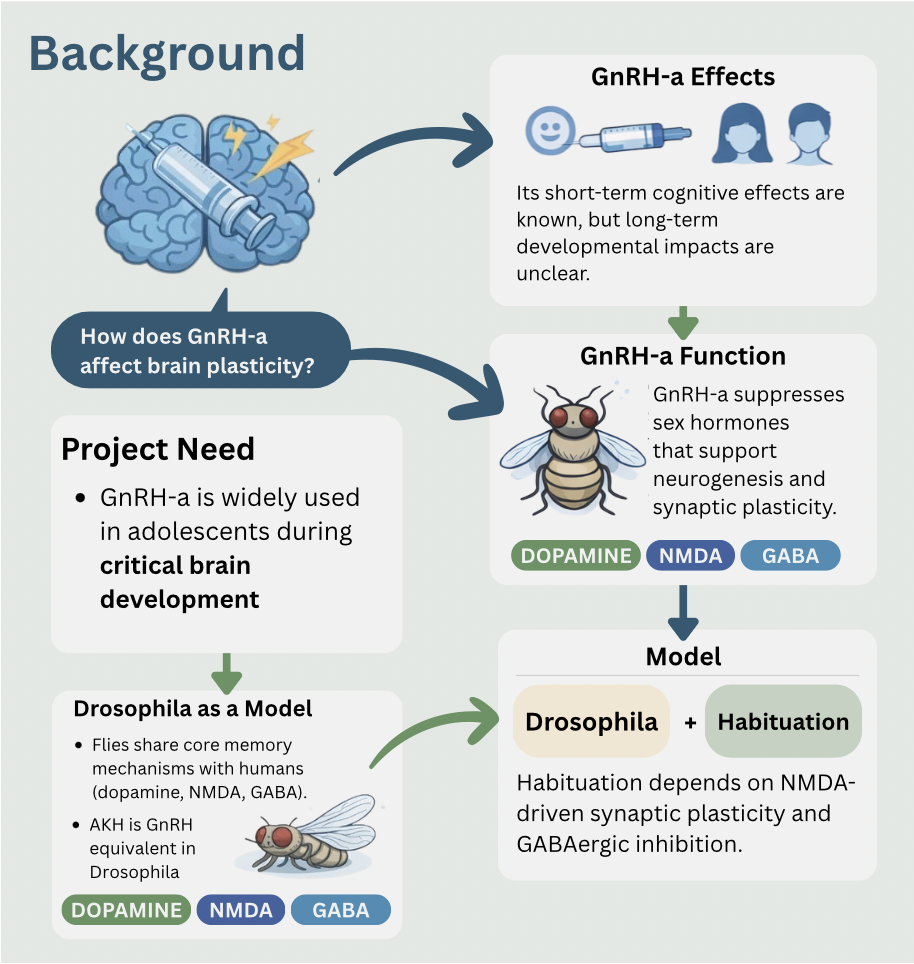
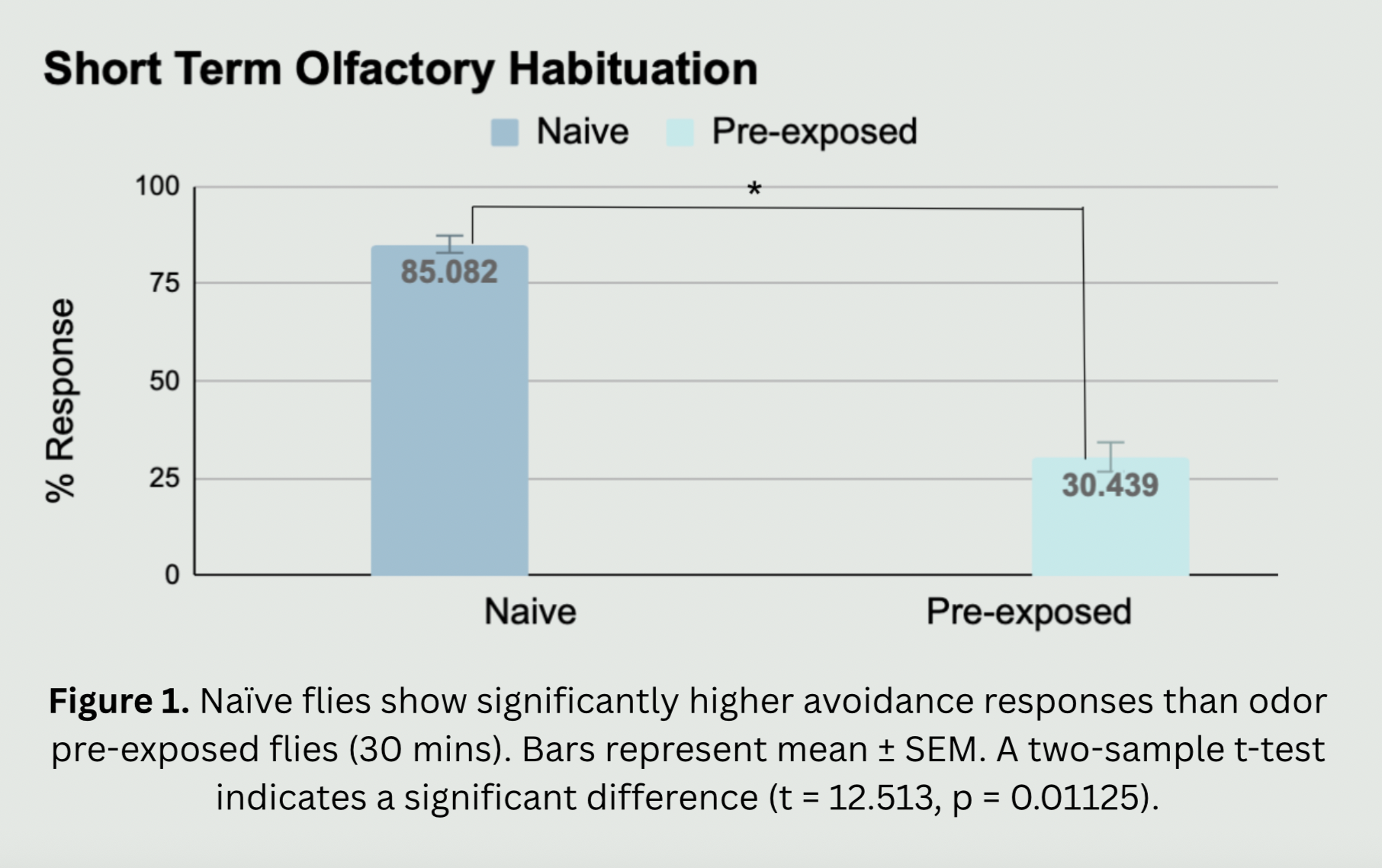
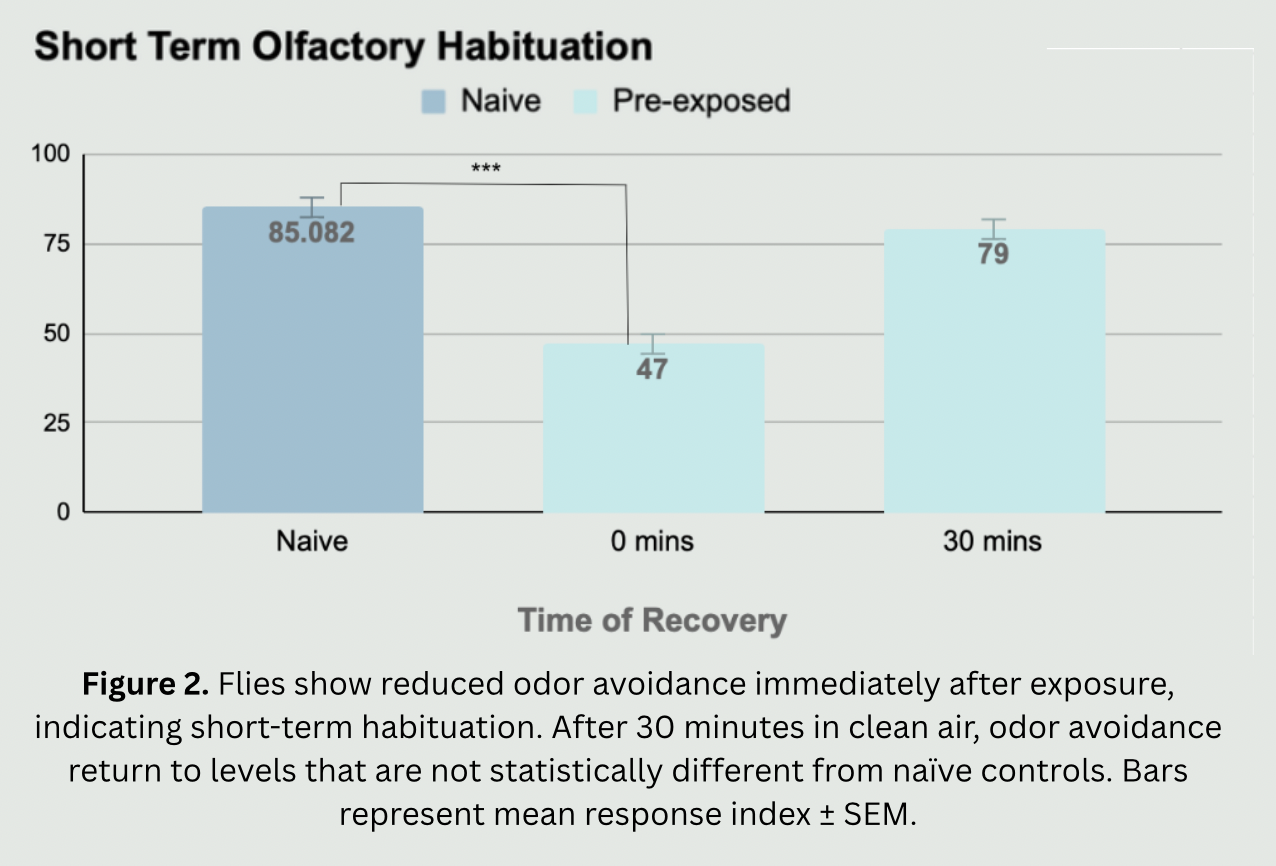
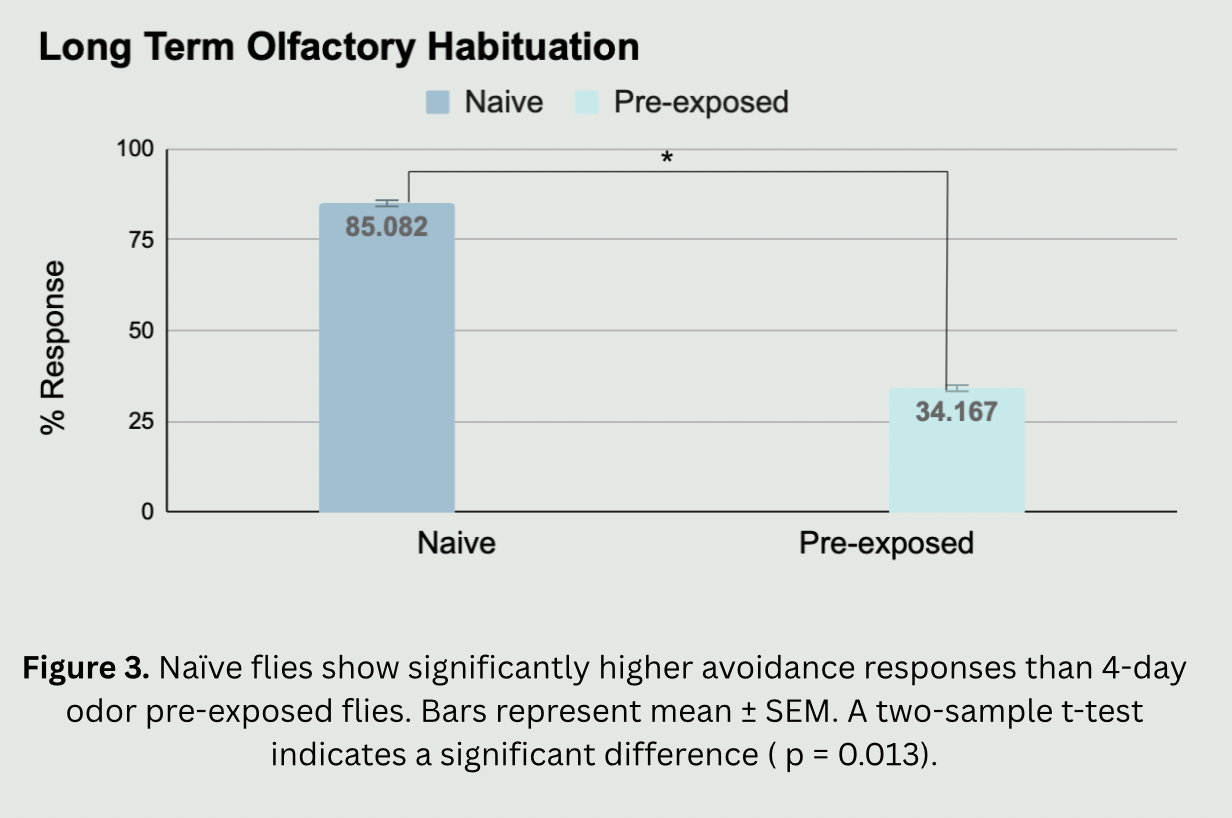
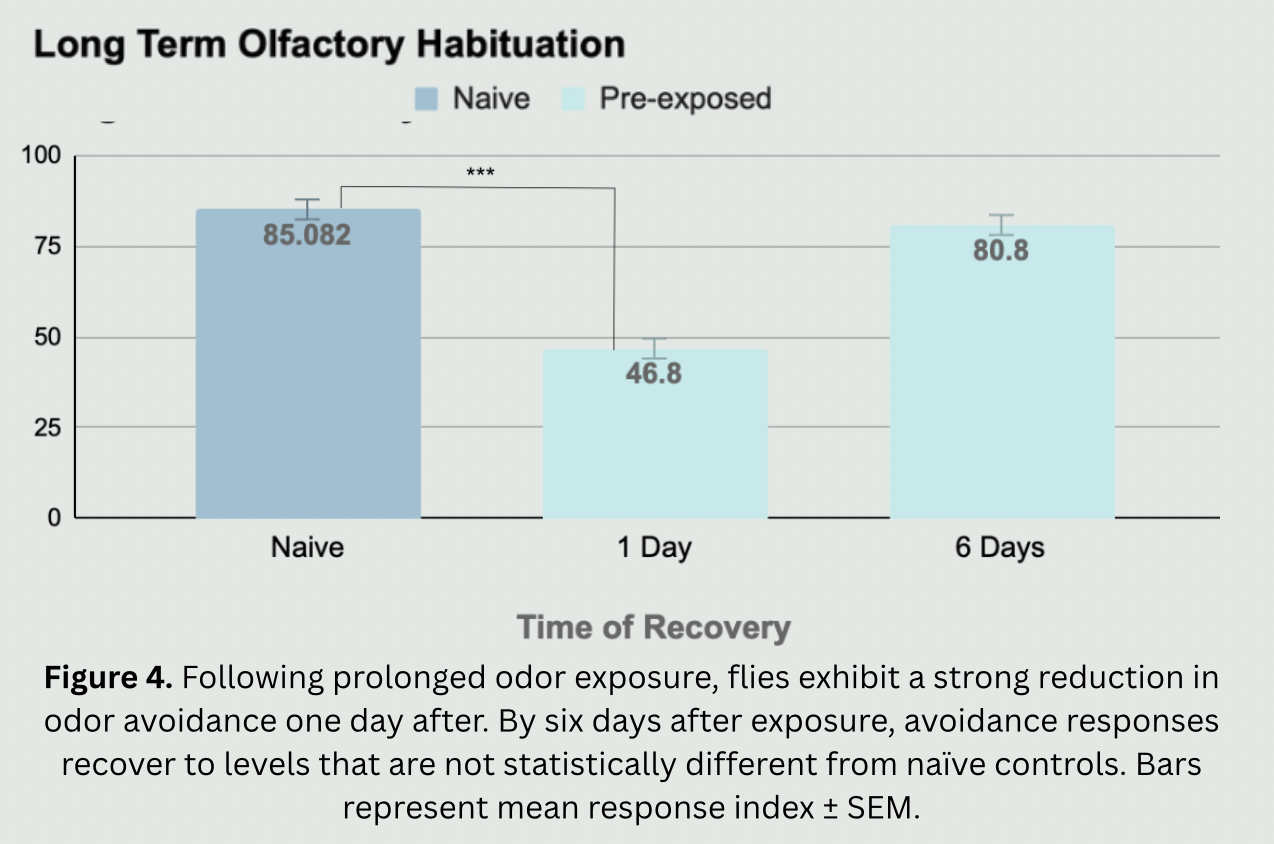
The need for this project stems from the
controversy regarding the widespread medical use of GnRH agonists.
Concerns are specifically for its use in children and teens
because as adolescents, they are still going through essential
brain development. Although this drug is extremely effective for
endometriosis, gender affirming care, and precocious puberty (the
delay of puberty in children who are developing too early) (Casati
et al., 2023), this treatment may have side effects on cognition.
One side effect of GnRH-a is short-term memory loss, which has
been shown through a study where perceived memory decreased
throughout subjects during treatment (Newton et al., 1996).
Despite widespread clinical use of GnRH agonists in pediatric
populations and researched short-term cognitive effects, there
remains limited mechanistic understanding of how hormonal
suppression during critical developmental periods could have long
term effects.
Gonadotropin-releasing hormone (GnRH) is mainly associated with
regulating the release of sex hormones through its action on the
hypothalamic pituitary axis. GnRH is produced in the hypothalamus,
where it to GnRH receptors and stimulates the release of sex
hormones such as estrogen and testosterone (Casati et al., 2023).
However, GnRH receptors are also found in the hippocampus and the
preoptic cortex parts of the brain which are essential for
cognition, suggesting that it has functions and effects that go
beyond reproduction.
In certain illness, cancer and illnesses that
rely on sex hormones, GnRH is a widespread treatment. The
treatment works by first overstimulating the pituitary gland and
gradually desensitizing the receptors and stopping the release of
gonadotropin hormones and sex hormones. This ultimately plays a
crucial role in our reproductive systems (Casati et al., 2023).
While this treatment is extremely affective, sex hormones that are
regulated by GnRH promote neurogenesis, regulates synaptic
plasticity, and memory formation (Kim & Casadesus, 2011; Celec et
al., 2015). As a result, GnRH is a factor in cognition and
suppressing GnRH indirectly alters the hormonal environment that
influences cognition.
If the suppression of GnRH occurs during a
developmental window when new neurons are forming and synapses are
being strengthened; it could have the ability to change how the
learning circuits originally form. Thus, the concern about GNRH-a
is both about how it affects memory during treatment as well as
whether temporary developmental disruption can lead to long
lasting changes even after hormone levels return to normal.
To investigate whether developmental
suppression of hormone signaling can result in lasting changes to
memory circuit, a genetically reliable and comparable model
organism is required. Drosophila melanogaster, a fruity fly, is
widely used to study how the brain learns because of its memory
mechanisms and processes that are similar than to those in humans.
Although the brain of Drosophila is smaller and less complex than
the human brain, it uses the same types of neurons and molecules
including dopamine for reinforcement learning, NMDA receptors for
synaptic plasticity, and GABAergic neurotransmitter for
inhibition, all of which are imperative to building and modifying
memory. Although drosophila does not possess the GnRH gene, it
still provides as a comparable model for this project because it
has a structural equivalent, the adipokine tic hormone (AKH)
(Beh-Manahem, 2021). While AKH is not genetically homologous to
GnRH, it serves as a functional endocrine analog that modulates
dopaminergic learning circuits in a comparable manner.
Flies show
a similar process to how GnRH affects cognition through AKH. AKH
is released into the blood by endocrine cells in the corpora
cardiaca (neuroendocrine glands that function similarly to the
vertebrate pituitary gland) and travels to neurons in the
suboesophageal zone that expresses AKH receptors. As the AKH binds
to these neurons, it alters how they activate dopaminergic neurons
and how much dopamine is released during learning (Meschi et al.,
2024). Recent studies showed that AKH can change how dopamine can
neurons respond during aversive learning which allowed how hunger
altered the memories of drosophila. Because dopamine driven
learning in flies depends on the synaptic plasticity in the
mushroom body, drosophila’s primary learning center, AKH’s
influence on dopamine affects the synaptic plasticity that is
required for memory.
NMDA (N-methyl-G-aspartic acid) receptors that are activated by
the NMDA molecule and is an essential factor of synaptic
plasticity. Synaptic plasticity is the process where the
connections of the neurons change as they get more experienced.
Because synaptic plasticity is how the neurons adapt to new
experiences or respond with repetition, it can be drawn into
memory formation and the process of habituation, a non-harmful,
repeated stimuli which results in decreased behavioral responses
(Paoletti et al., 2013).
Habituation uses non stressful stimulus
and directly reflects synaptic plasticity. Essentially,
habituation reflects the maturation and flexibility of the neural
circuits. As such, habituation provides as a sensitive behavior
for detecting subtle disruptions in synaptic plasticity. In
Drosophila, habituation depends on synaptic plasticity which is
influenced by NMDA receptors and GABAergic interneurons that
lessen neural responses over time (Larkin et al., 2010; Das et
al., 2024). Human mechanisms also use the NMDA plasticity and
GABAergic inhibition. Habituation is also a better choice than
aversive learning tests for this project due to the fact that
aversive conditioning requires electric shock, which activates
reactions like stress, pain, etc. Because there are so many
factors that go into aversive learning it is more difficult to
observe if any differences are due to memory circuits or other
systems. Therefore, examining habituation learning in Drosophila
following developmental AKH suppression provides an approach to
isolating long-term effects on memory related plasticity.