Quad Chart
If you are unable to view the file, click here.
The Effect of Transgenerational Epilepsy on Dementia Pathology
This study investigates whether epilepsy can cause transgenerational cognitive impairments in Caenorhabditis elegans. Seizures were induced using PTZ, and spatial learning was assessed across three generations. Results show that even a single seizure exposure can impair learning in descendants — suggesting that seizure-induced epigenetic changes may be inherited and could link epilepsy to increased dementia risk across generations.
→ View the STEM I Subpage (Grant, Notes, Video)
Abstract
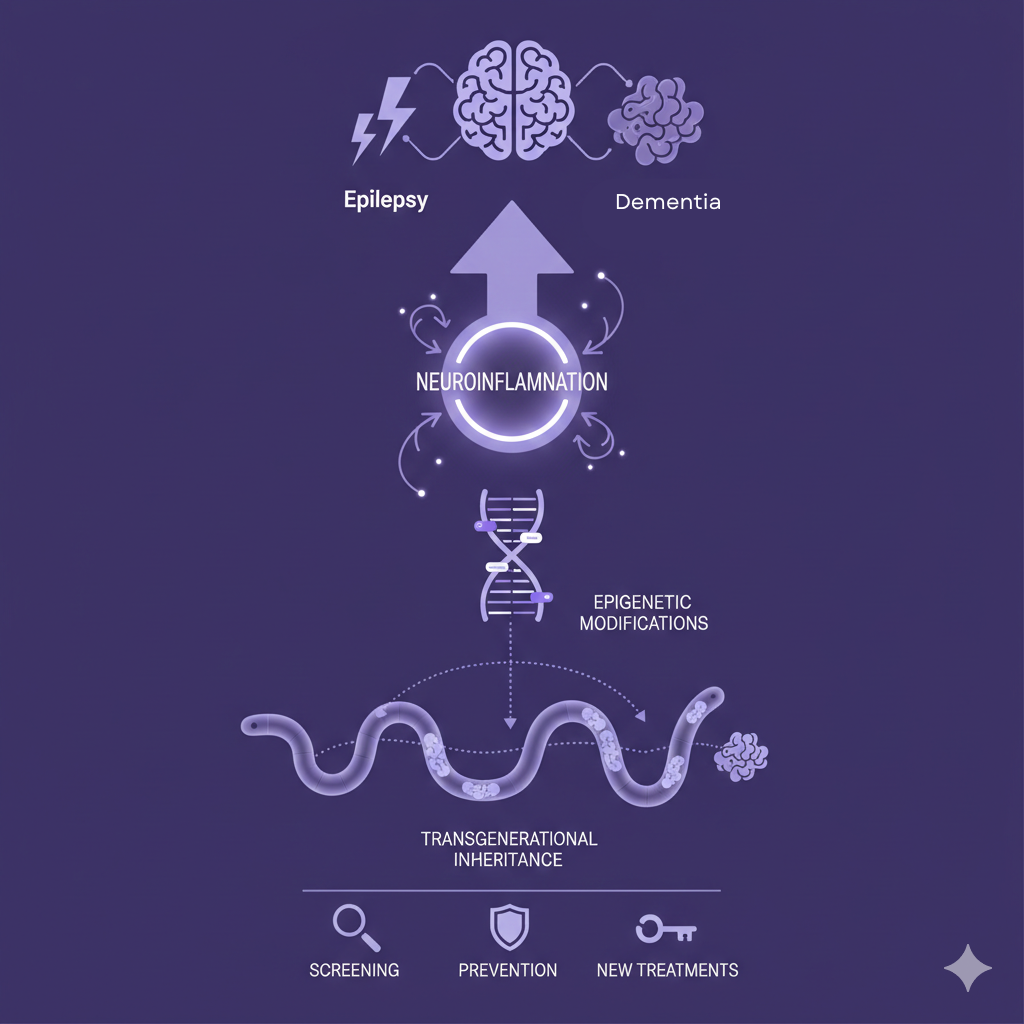
Epilepsy and Alzheimer's disease are increasingly recognized as comorbid neurological disorders, yet the mechanisms linking them remain unclear. Seizure-induced neuroinflammation may drive epigenetic changes that alter gene expression and can be inherited across generations, potentially increasing dementia risk.
This study investigates whether epilepsy produces transgenerational cognitive impairments using C. elegans as a model organism. Seizures were chemically induced using pentylenetetrazole (PTZ). Spatial learning was assessed across three generations using a T-maze chemotaxis assay, comparing exposed worms to unexposed controls.
Control worms consistently showed robust spatial learning (p < 0.0001). PTZ exposure impaired learning in a dose- and generation-dependent manner. In Generation 3, cumulative exposure further exacerbated impairments, with repeatedly exposed worms failing to improve during testing (p > 0.05). Learning deficits persisted after PTZ was discontinued, indicating lasting inherited effects.
Keywords: Epilepsy, Alzheimer's, dementia, epigenetics, transgenerational inheritance, C. elegans
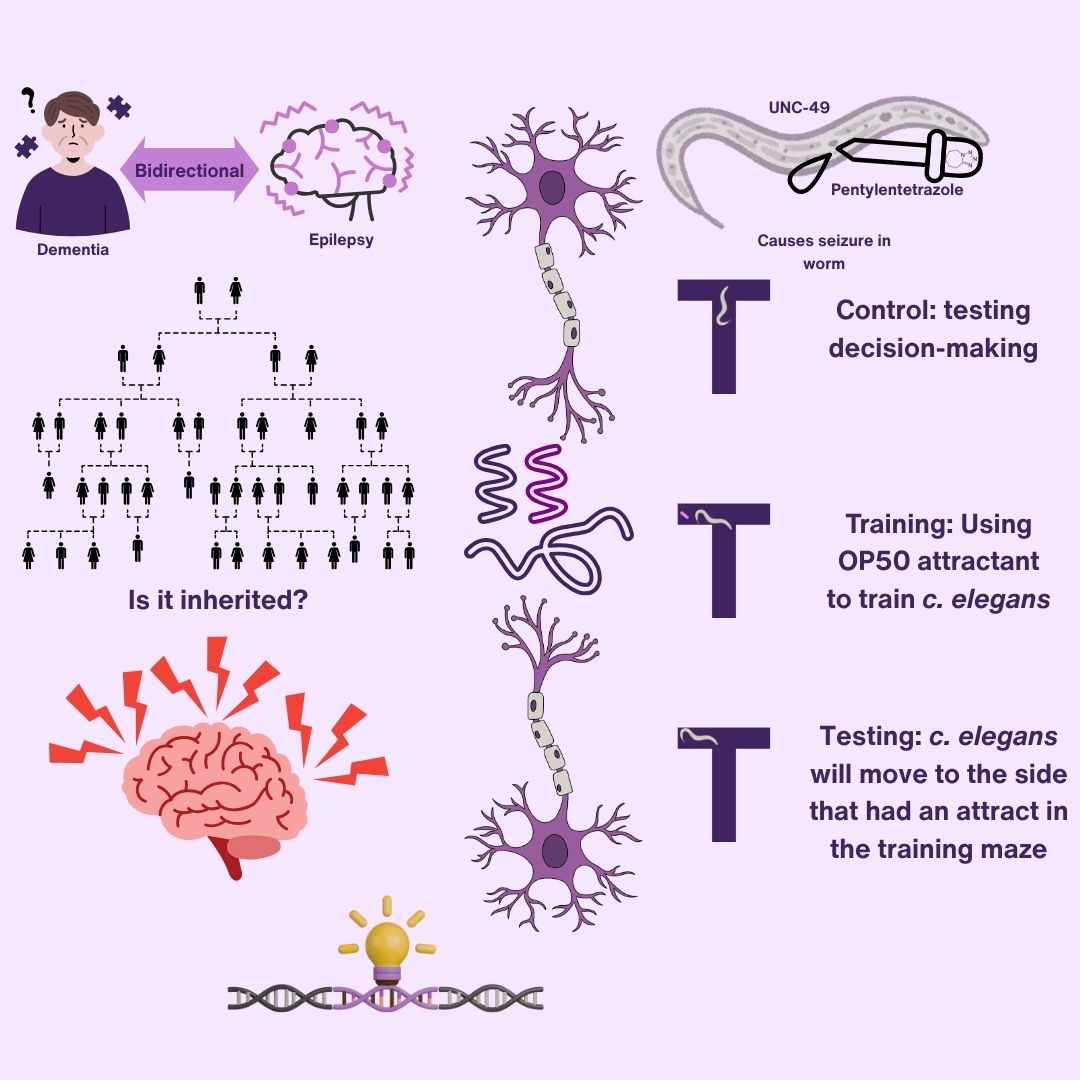
Research Question
How does dementia pathology in C. elegans change due to epilepsy over many generations?
Hypothesis
If more generations of C. elegans are given seizures, then future generations will have more dementia pathology because of inherited epigenetic modifications.
Background
Epilepsy and Alzheimer's disease (AD) are highly prevalent neurological disorders affecting over 50 million people annually (Yang et al., 2022). Research increasingly supports a bidirectional relationship between them, driven by shared neuroimmune and neuroinflammatory pathways (Stewart & Johnson, 2025). Epilepsy involves recurrent seizures caused by genetic and environmental factors, with many cases linked to structural or unknown etiologies (Zhang et al., 2024). Chronic neuroinflammation in epilepsy can trigger epigenetic modifications, including altered gene expression and DNA methylation (Komada & Nishimura, 2022), which are implicated in AD pathogenesis (Sharma et al., 2020). Because epigenetic changes can be transgenerational (Fitz-James & Cavalli, 2022), epilepsy-related molecular alterations may increase AD risk across generations.
C. elegans serves as an effective model organism for studying these mechanisms due to its mapped connectome, genetic simplicity, and established use in epilepsy and AD research (Emmons et al., 2021; Gourgou et al., 2021).
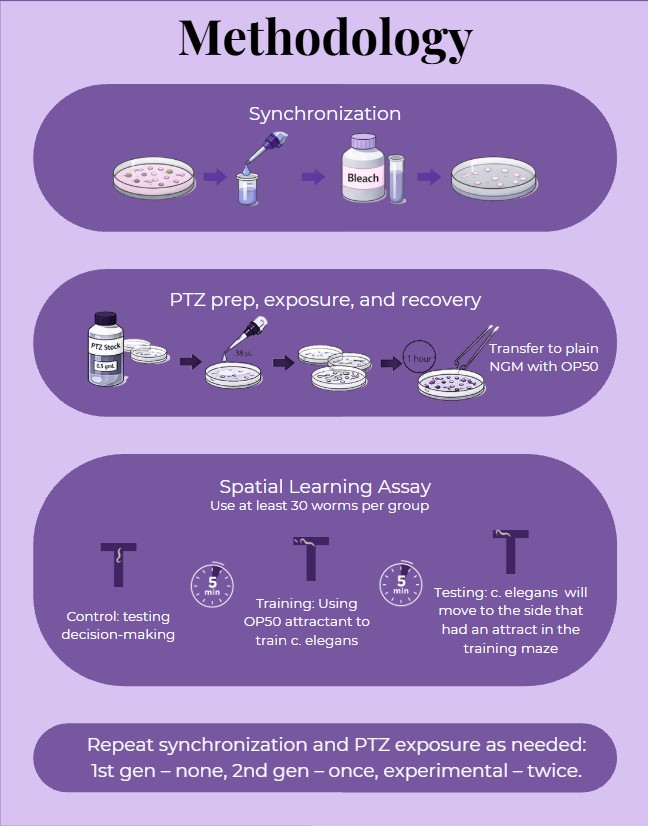
Procedure
Equipment & Materials
This study utilized C. elegans unc-49 worms on NGM agar plates seeded with E. coli OP50. Age-synchronized populations were obtained using a standard bleach protocol. PTZ exposure plates were prepared using a 0.5 g/mL stock solution on 100 mm NGM plates.
C. elegans Synchronization
Worms were synchronized using bleach treatment and divided into four groups: Control, 1-generation PTZ, 2-generation PTZ, and 3-generation PTZ. They were allowed to develop for two days before exposure to ensure consistent larval stage across groups.
PTZ Exposure
Thirty synchronized worms were transferred to PTZ-treated plates for 10–15 minutes, then allowed to recover on standard NGM plates for at least one hour before behavioral testing. This enabled assessment of neural excitability and cumulative effects across generations.
T-Maze Assay
Individual worms were placed at the start of a T-maze, with one arm containing OP50 as a food reward during training. Worm distribution was recorded after a 5-minute training phase, a second trial, and finally on an empty maze to assess memory without food cues.
Statistical Analysis
Z-proportion tests compared the observed proportion of worms selecting the conditioned arm to the expected probability by chance (0.5), providing a framework for evaluating whether choices were non-random and significantly influenced by treatment.
Results
Unable to view? Open PDF
Analysis & Discussion
Unable to view? Open PDF
Conclusion
Unable to view? Open PDF
References
Unable to view? Open PDF
Poster
Unable to view? Open PDF