STEM 1, taught by Dr. Crowthers, mainly focuses on helping us develop and conduct our independent research projects. Over the summer, we began brainstorming potential project ideas through pie charts and mind maps, and then during the first few weeks of school, we finalized our project ideas and began working on them. During the months of October to December, we collect preliminary data and analysis, and we present our projects at December fair, a mock version of February fair. After December fair, we collect our primary data and analyze it, which is presented at February Fair. 12 students at February fair are selected to advance to WRSEF, and then MSEF and onwards. This class consists of a lot of in-class work time for the project and teaches students to manage their time effectively. Each term, we have an informal meeting with no grade, as well as a formal meeting where we present our project or an article to Dr. C. The second half of STEM is STEM 2. Check out that page for more information! Trouble viewing the PDF? Click here!
Overview: This project studies how different levels of electrical stimulation affect the action potentials of a neuromodulated cockroach leg. The results found indicate that there is an optimal stimulation window of electrical stimulation that reduces excitotoxic activity in glutamate treated motor nerves.
Amyotrophic Lateral Sclerosis (ALS) is a neurodegenerative disease that targets motor neurons in humans and ultimately causes cell death. Annually, over 5,000 people die of ALS, as there is currently no cure (Centers for Disease Control and Prevention [CDC], 2024). In ALS patients, glutamate is not cleared properly due to dysfunction of the EAAT2 glutamate transporter. The resulting buildup of glutamate and increased calcium influx in motor neurons eventually leads to excitotoxic damage and motor neuron death (Le Gall et al., 2020). Although scientists have identified how glutamate increases calcium influx through NMDA receptors and contributes to excitotoxicity in ALS, the relationship between glutamate-induced hyperexcitability and electrical stimulation has not been fully understood (Spreux-Varoquaux et al., 2002).
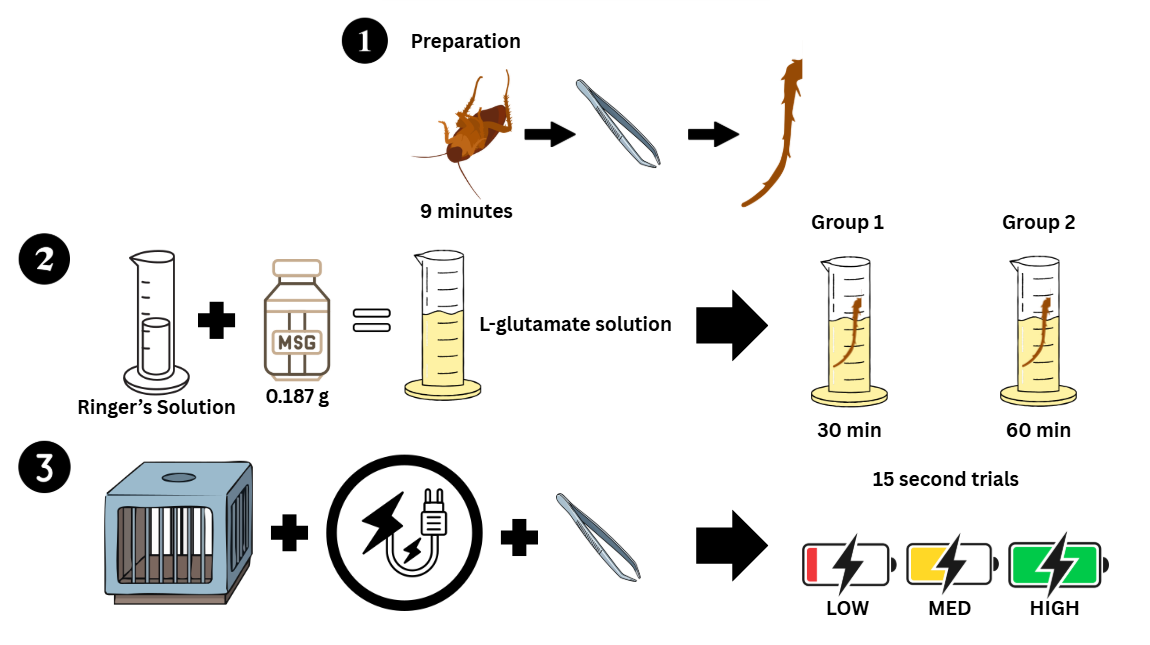
Using Periplaneta americana cockroaches, this project aims to quantify the relationship between neuronal excitability and electrical stimulation under glutamate-induced conditions. The methodology consisted of bathing a cockroach leg in L-glutamate to simulate an ALS-like environment, applying three levels of electrical stimulation (low, medium, and high), and recording action potentials using a Backyard Brains SpikerBox. Data was collected from cockroach legs exposed to L-glutamate for 30 minutes and 60 minutes to represent different stages of excitotoxic stress.
Results showed that in the 30-minute data sample, low-frequency electrical stimulation helped keep neurons active. In the 60-minute sample, low-frequency stimulation protected motor neurons as they became excitotoxic. Overall, the data suggests the existence of a stimulation “sweet spot,” in which low electrical stimulation may support neuronal stability and potentially reduce excitotoxicity in an ALS-like environment.
Keywords: ALS, motor neuron, glutamate, calcium, NMDA, EAAT2

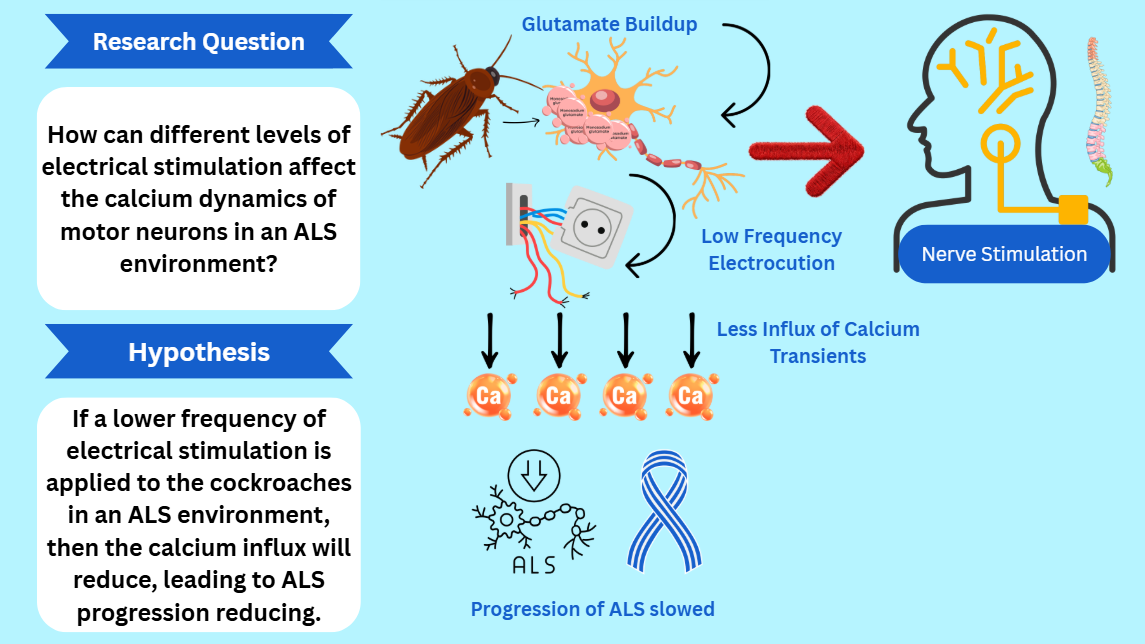
How do different frequencies of electrical current affect the action potential of cockroach motor neurons under different exposure levels to L-glutamate?
If a lower frequency of electrical current is applied to the neuromodulated cockroach, this will result in the largest decrease in intercellular calcium levels in all exposure time periods, because in a normal system, lower frequencies of electrical current lead to less neural activity and less glutamate release so calcium levels do not rise. This hypothesis will be displayed by lower action potential amplitudes with and without hair stimulation.
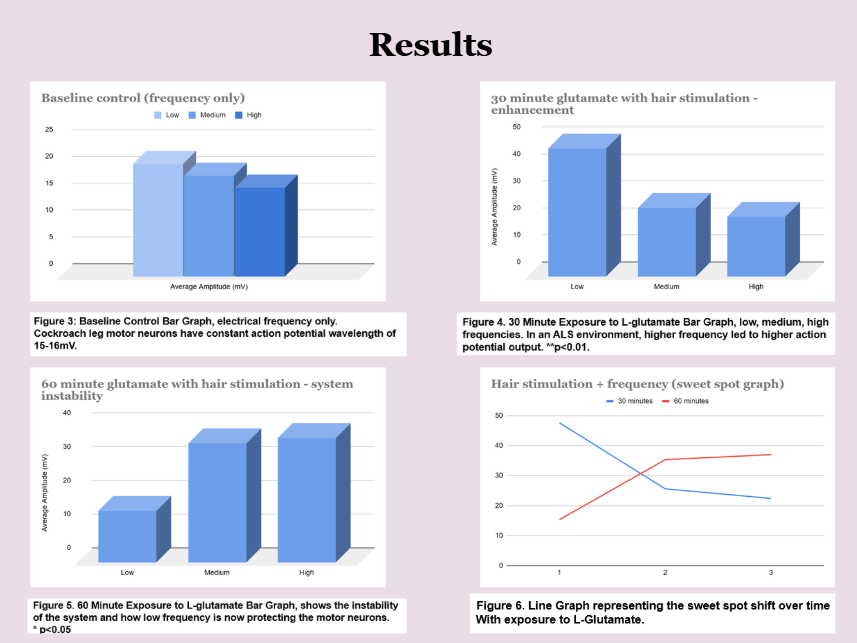
The results found that neuronal response exhibits a frequency “sweet spot” where activity is strong but remains stable, avoiding calcium overload and excitotoxic stress. In control nerves, the sweet spot is broad, indicating a robust system tolerant to stimulation changes. After short glutamate exposure (30 minute), low-frequency stimulation produces the largest responses by allowing recovery between spikes. After prolonged glutamate exposure (60 minute), the optimal window shifts, reflecting increased fragility and excitotoxic vulnerability. Low-frequency stimulation transitions from optimal performance early to protective stabilization later, rather than being universally “best.” Lower electrical frequencies can help lower excitotoxic stress in ALS-like conditions.