Fostering intellectual curiosity, the STEM class guides students through the science fair and research process regardless of prior experience. Through project-based learning, mentorship, and hands-on experimentation, students learn how to develop original research questions, design and conduct experiments, analyze data, and clearly present their findings. The class encourages students to think independently, troubleshoot challenges, and refine their ideas through trial and error. By emphasizing critical thinking, creativity, and real-world problem solving, the course builds both confidence and technical skills, preparing students for competitive science fairs and future STEM endeavors.
Project: Using mediated drug therapy to disrupt SLC6A4 function in adult C. elegans to study how hereditary serotonin transporter deficits influence molecular and behavioral markers linked to hereditary suicide vulnerability in adolescent C. elegans. I hypothesize that if the serotonin signaling molecules are increased in adult parental organisms, then their offspring will show decreased levels of suicidal inclinations with anti-depressant medication because this is similar to how SSRIs work to prevent SERT re-uptake of serotonin signaling molecules which promote an increase in serotonin signaling molecules.

Using mediated drug therapy to selectively disrupt SLC6A4 (serotonin transporter) function in adult C. elegans, this study aims to investigate how heritable deficits in serotonin reuptake affect molecular signaling pathways, neurotransmitter balance, and behavioral phenotypes associated with suicide vulnerability in adolescent offspring. By analyzing changes in serotonin-dependent gene expression, neural circuit activity, and stress-related or risk-associated behaviors, we aim to elucidate the mechanistic link between parental serotonergic modulation and intergenerational transmission of susceptibility to depressive- and suicidal-like phenotypes, providing insight into fundamental biological mechanisms underlying hereditary mood disorders.
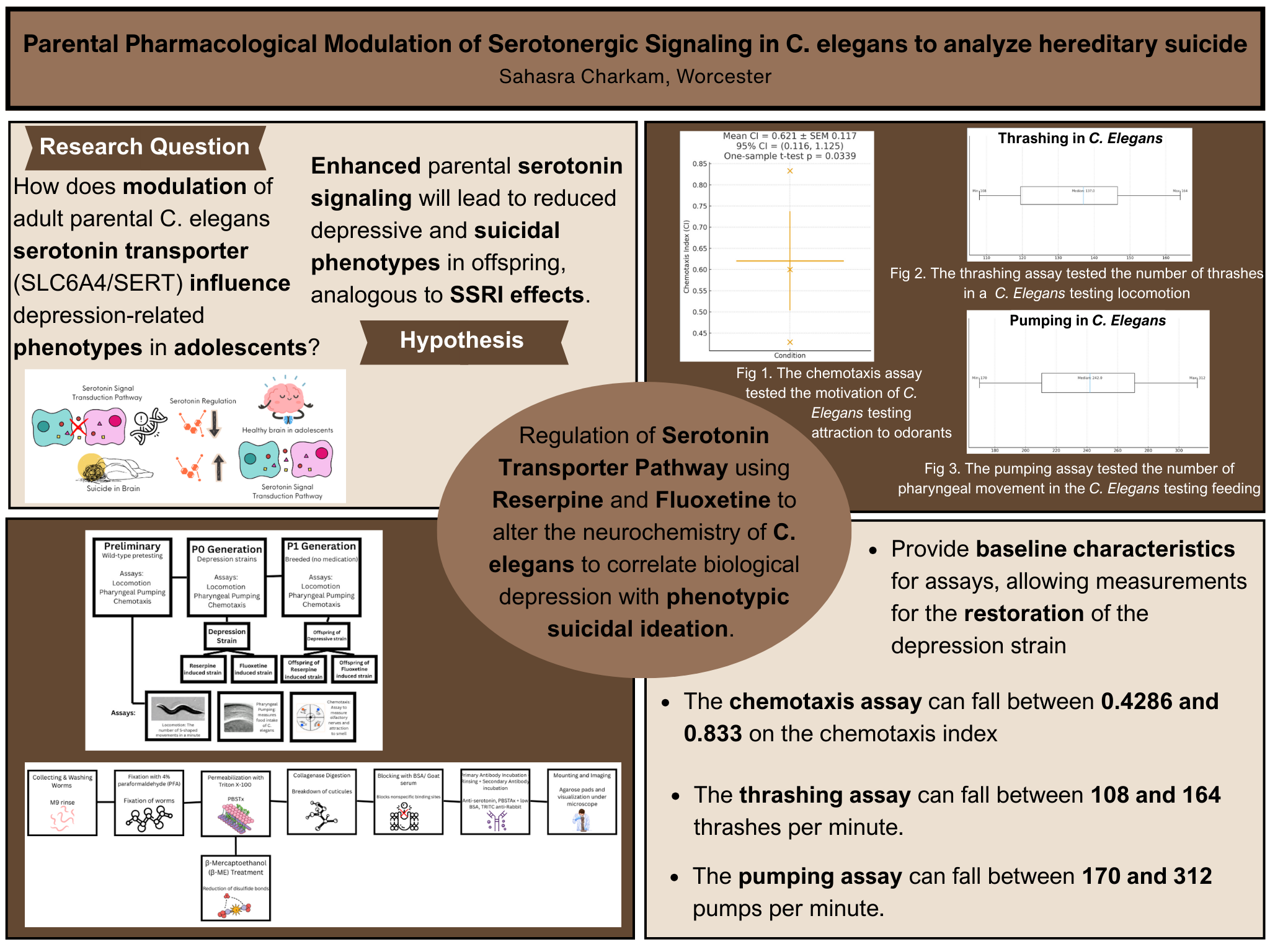
Suicide runs through familial genetics, putting children at risk for suicide and depression, primarily based on biological characteristics. One of these biological influences is the SERT transporter gene, which in turn impacts the serotonin signaling. Serotonin signaling broadly modulates neural circuits involved in emotional processing, behavioral regulation, and stress responsiveness, thereby influencing mood stability and vulnerability to affective disorders (Cowen and Browning, 2015). This project questions how the alteration of adult parental C. elegans serotonin transporter (SLC6A4/SERT) impacts the behavioral and biological presence of suicidal ideation in adolescents. C. elegans behavior parallels that of humans through feeding, locomotion, and motivational attraction reactions, proving to be a viable model organism for this project. Inducing the organisms with reserpine and fluoxetine, the serotonin signaling will be downregulated and upregulated to test their behavior during the P0 generation. To analyze the hereditary behavior of the F1 generation will also be collected to draw a correlation between serotonin regulation and heredity. The pumping assay showed that P0 worms treated with 6.25 µM reserpine exhibited reduced rates of approximately 150 pumps/min compared to wild-type controls at ~250 pumps/min, while F1 offspring of fluoxetine-treated (6.25 µM) parents displayed restored pumping rates of approximately 255–265 pumps/min, comparable to control levels, demonstrating rescue in the offspring. The project aimed to prove a correlation of suicidal heredity and that if the parent is treated with a rescue drug, the offspring will present with normal symptoms.
Visual Summary: The experimental design shows wild-type C. elegans exposed to two doses of reserpine (6.25 µM and 1.25 µM) to induce serotonin depletion and depression-like phenotypes. After 24 hours, organisms were divided into three groups: continued reserpine exposure, fluoxetine rescue treatment, or standard conditions. Behavioral assays (thrashing and pharyngeal pumping) were conducted on the parental (P0) generation at 24 and 48 hours. F1 offspring from each treatment group were raised without drug exposure and assessed for transgenerational behavioral effects. Results demonstrated dose-dependent parental impairment with differential fluoxetine rescue efficacy, and remarkable transgenerational recovery in offspring, particularly in the low-dose treatment group.
Click here to view the full research proposal and project details
Research Question: How does the alteration of adult parental C. elegans serotonin transporter (SLC6A4/SERT) impact the behavioral and biological presence of suicidal ideation in adolescents?
Initial Hypothesis: If parental C. elegans experience pharmacologically induced serotonin dysregulation, then their offspring will exhibit altered behavioral phenotypes associated with depression-like states, as serotonergic dysfunction is transmitted through epigenetic or developmental mechanisms.
Refined Hypothesis: If adult parental Caenorhabditis elegans experience decreased serotonin transporter (SLC6A4/SERT) activity through reserpine treatment, then their offspring will exhibit transgenerational behavioral recovery when parents receive fluoxetine rescue treatment, due to restoration of parental serotonergic signaling during critical periods of germline development. Furthermore, moderate parental serotonergic disruption will permit more complete transgenerational compensation than severe disruption, suggesting threshold effects in developmental buffering mechanisms.
Hereditary Suicide Risk: Fifty-five percent of suicide risk is heritable, independent of psychiatric disorders (Voracek and Loibl, 2007). Offspring of parents with suicidal behaviors exhibit increased biological vulnerability, yet relatively few studies have targeted the biological pathways of hereditary suicidal ideation. Suicide risk is polygenetic and strongly associated with Major Depressive Disorder (MDD), which increases the likelihood of suicidal ideation—the thought of ending one's life.
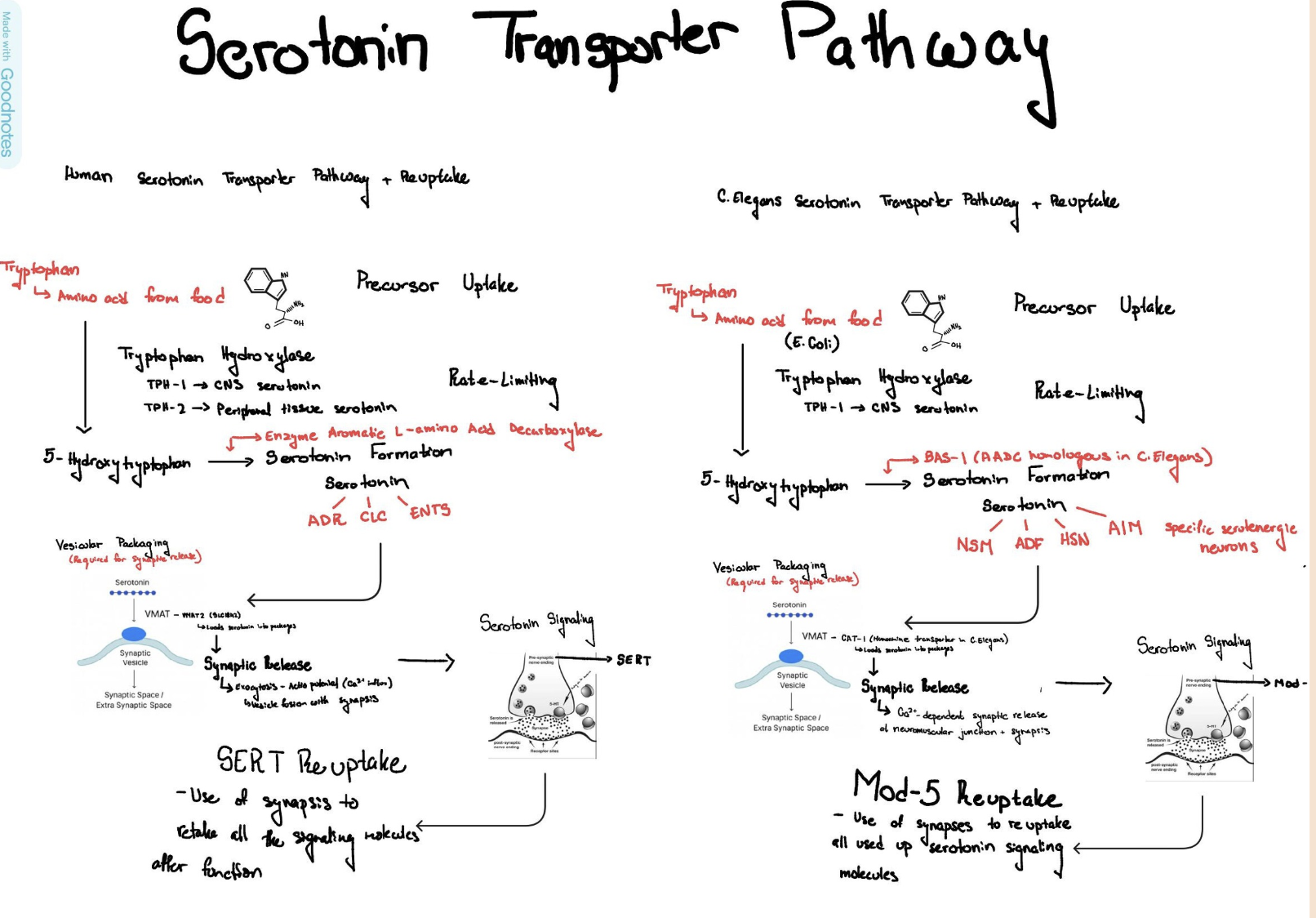
Serotonergic Dysfunction: One key biological influence is the serotonin transporter (SERT) gene, which encodes the SLC6A4 protein responsible for serotonin reuptake. Serotonin signaling modulates neural circuits involved in emotional processing, behavioral regulation, and stress responsiveness, thereby influencing mood stability and vulnerability to affective disorders (Cowen and Browning, 2015). Disruptions in serotonergic signaling have been consistently implicated in depression and suicide risk.
C. elegans as a Model: Caenorhabditis elegans provides a powerful model for studying serotonergic mechanisms due to its completely mapped nervous system (302 neurons), sequenced genome, and conserved serotonin transporter gene (mod-5), which is functionally equivalent to human SLC6A4. Despite anatomical simplicity, C. elegans exhibit complex behaviors—feeding, locomotion, chemotaxis, and arousal—all modulated by serotonin. The transparent body allows live neuronal imaging, and the short generation time (3 days) enables cross-generational studies of inherited neurochemical changes. This makes C. elegans an ideal organism for investigating how parental serotonergic dysfunction influences offspring behavior across generations.
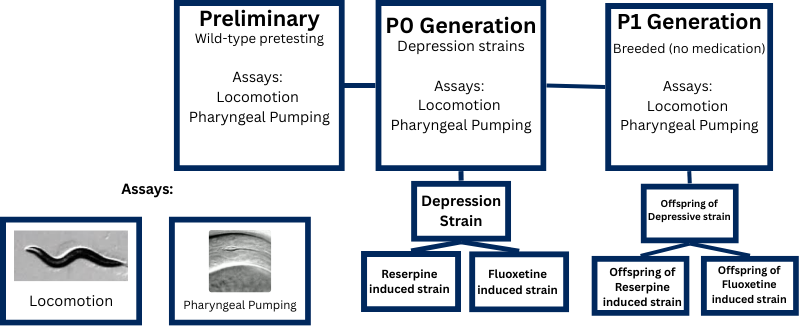
Synchronization: A synchronized population of Caenorhabditis elegans was established using standard alkaline hypochlorite bleaching protocol (Massachusetts Institute of Technology, 2015) to ensure all experimental organisms were at the same developmental stage. Eggs were collected and allowed to hatch on NGM plates seeded with OP50 bacteria.
Baseline Assessment: Wild-type (N2 strain) worms were assessed for baseline behavioral parameters using thrashing assays (body bends per minute in M9 buffer) and pharyngeal pumping assays (pumps per minute observed under dissecting microscope). Wild-type baseline values were established as 136.8 ± 14.6 thrashes/min and 256.6 ± 19.7 pumps/min (N=10 for each assay).
Pharmacological Treatment: Synchronized young adult worms were exposed to reserpine (a serotonin-depleting agent that inhibits vesicular storage) at two concentrations: 6.25 µM (high dose) and 1.25 µM (low dose). After 24 hours of reserpine exposure, organisms were divided into three treatment groups: (1) continued reserpine exposure for an additional 24 hours, (2) fluoxetine rescue treatment (100 µM) for 24 hours to restore serotonergic signaling, or (3) 24-hour reserpine-only control.
P0 Behavioral Assays: Parental generation (P0) organisms were assessed at 24 hours (after initial reserpine treatment) and 48 hours (after rescue or continued treatment) using thrashing and pharyngeal pumping assays. Ten organisms per treatment group were tested, with behavioral measurements recorded for statistical analysis.
F1 Generation Assessment: Treated P0 worms were transferred to fresh NGM plates and allowed to lay eggs. F1 offspring developed without any pharmacological exposure. Once F1 organisms reached young adulthood, they underwent the same behavioral assays (thrashing and pumping) to evaluate transgenerational effects of parental serotonergic modulation.
Data Collection and Analysis: All behavioral data were recorded and analyzed using Student's t-tests (two-tailed, α = 0.05) to assess statistical significance. Comparisons included: treatment groups versus wild-type baseline (one-sample t-tests) and between different treatment conditions (independent two-sample t-tests). Results were graphed to visualize dose-dependent effects, rescue efficacy, and transgenerational recovery patterns.
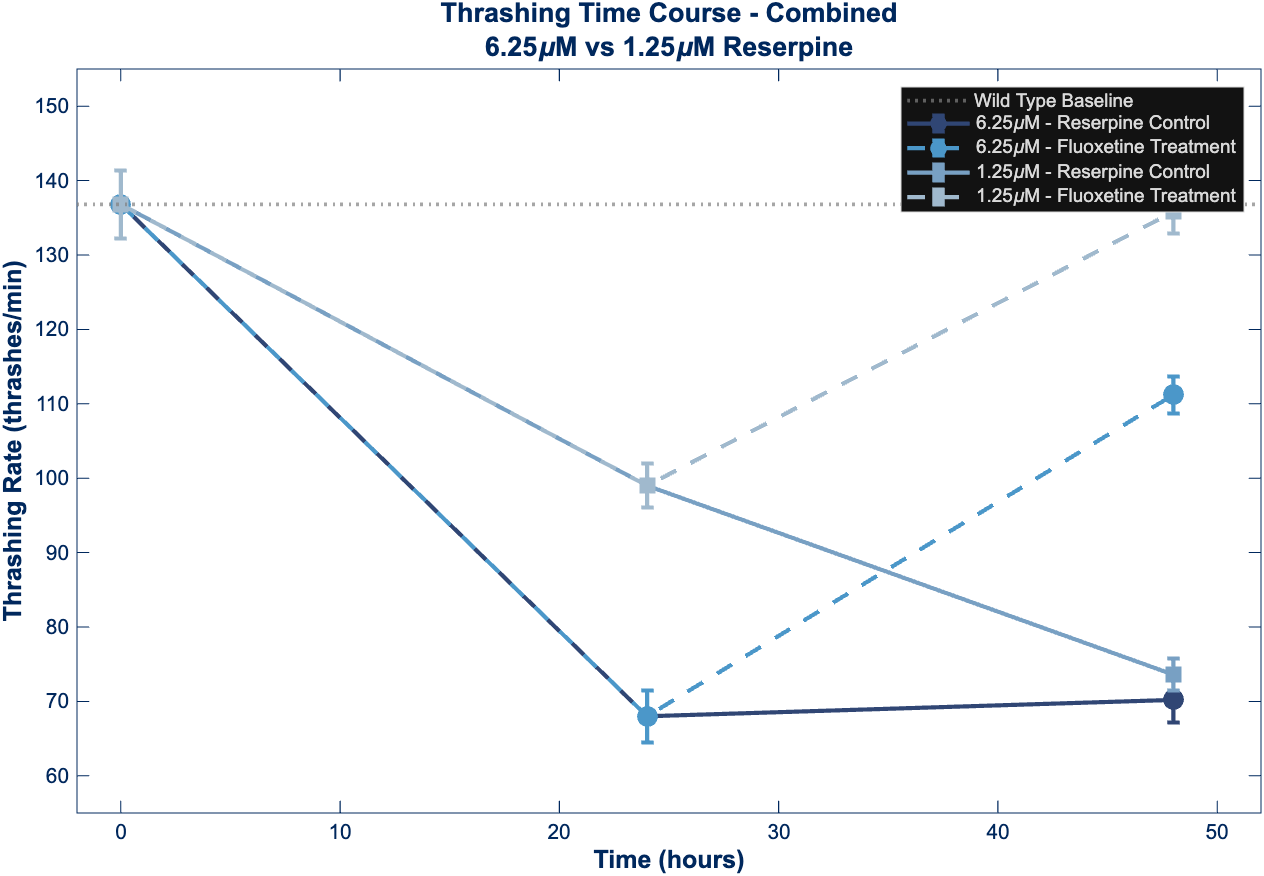 Figure 1 – Thrashing Time Course
Figure 1 – Thrashing Time Course
Time course analysis comparing thrashing behavior across 6.25 µM and 1.25 µM reserpine treatment groups. Both doses show initial decline in locomotor activity from wild-type baseline (dotted line at 136.8 thrashes/min). Fluoxetine treatment (dashed lines) produces dose-dependent recovery, with the 1.25 µM group achieving near-complete restoration (99.3% of baseline) compared to partial recovery in the 6.25 µM group (81.3% of baseline). Continued reserpine exposure (solid lines) results in persistent behavioral depression.
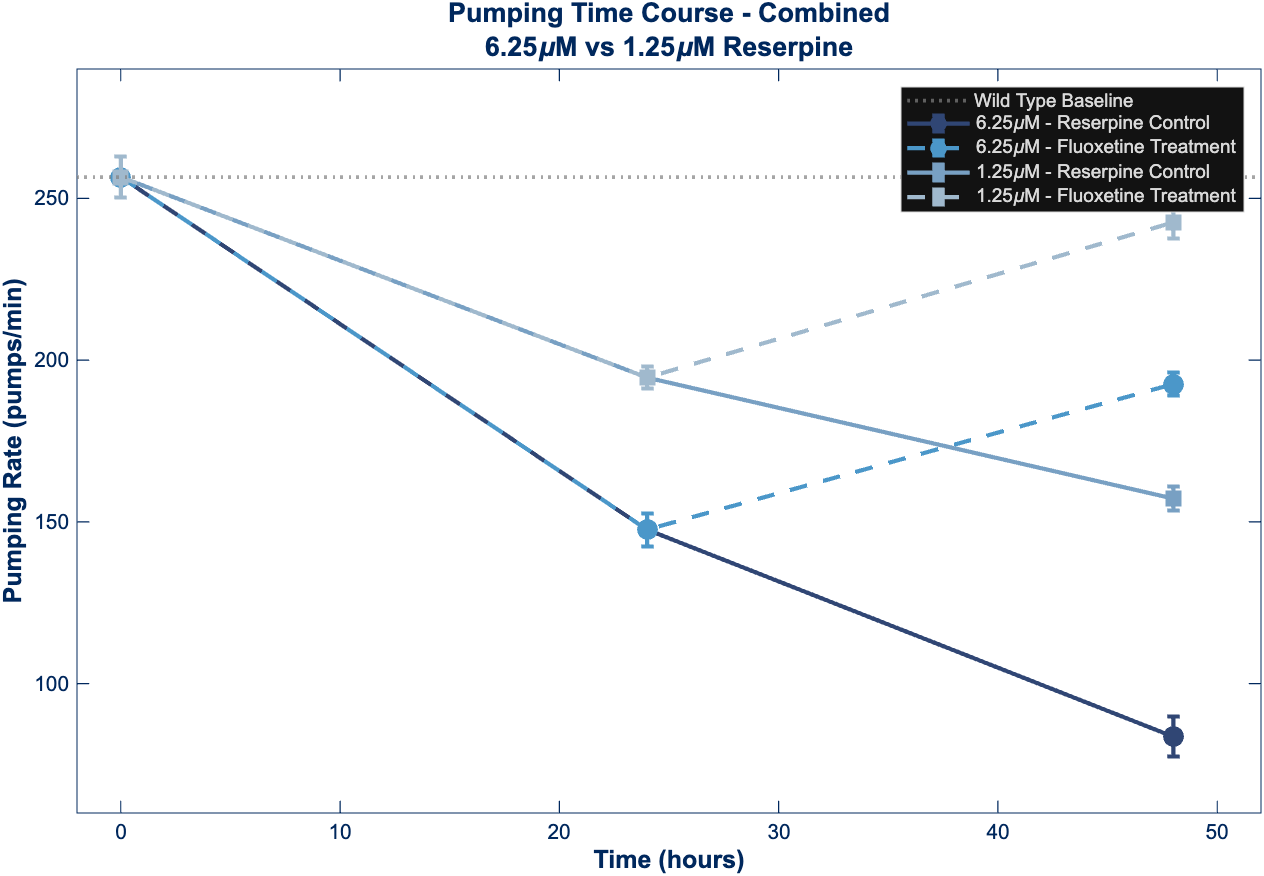 Figure 2 – Pharyngeal Pumping Time Course
Figure 2 – Pharyngeal Pumping Time Course
Feeding behavior time course showing dose-dependent impairment patterns similar to locomotion. The 6.25 µM reserpine group exhibits severe feeding deficits with limited fluoxetine rescue effectiveness (58.4% of baseline), while the 1.25 µM group demonstrates more robust recovery (93.0% of baseline). This differential rescue pattern suggests threshold effects where severe serotonergic disruption produces feeding deficits less responsive to acute SSRI intervention.
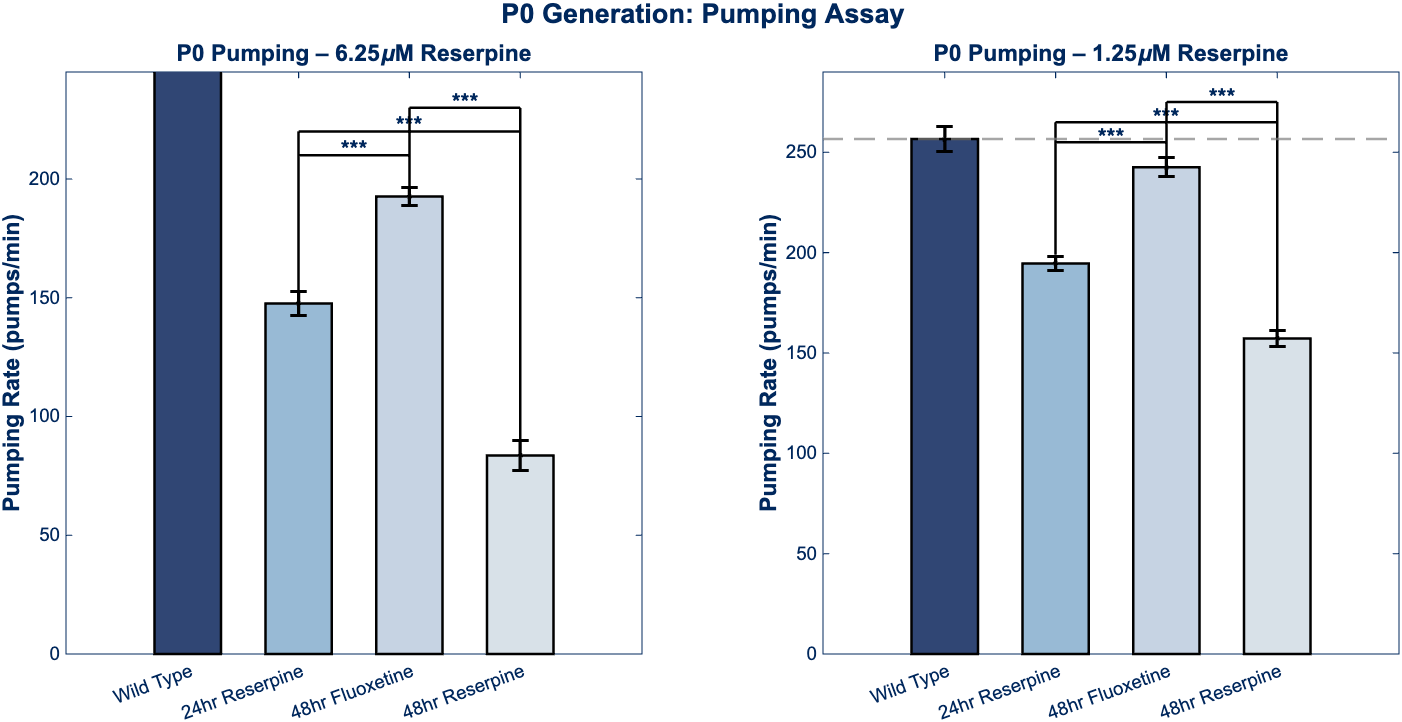 Figure 3 – P0 Generation Behavioral Assays
Figure 3 – P0 Generation Behavioral Assays
Comparative bar graphs showing P0 thrashing and pumping behavior for both dosage groups. Statistical significance (***p < 0.001) indicated for all comparisons between wild-type and reserpine-treated groups, and between reserpine and fluoxetine rescue conditions. The 6.25 µM reserpine treatment produces 50% reduction in thrashing and 43% reduction in pumping, while 1.25 µM produces 28% and 24% reductions, respectively.
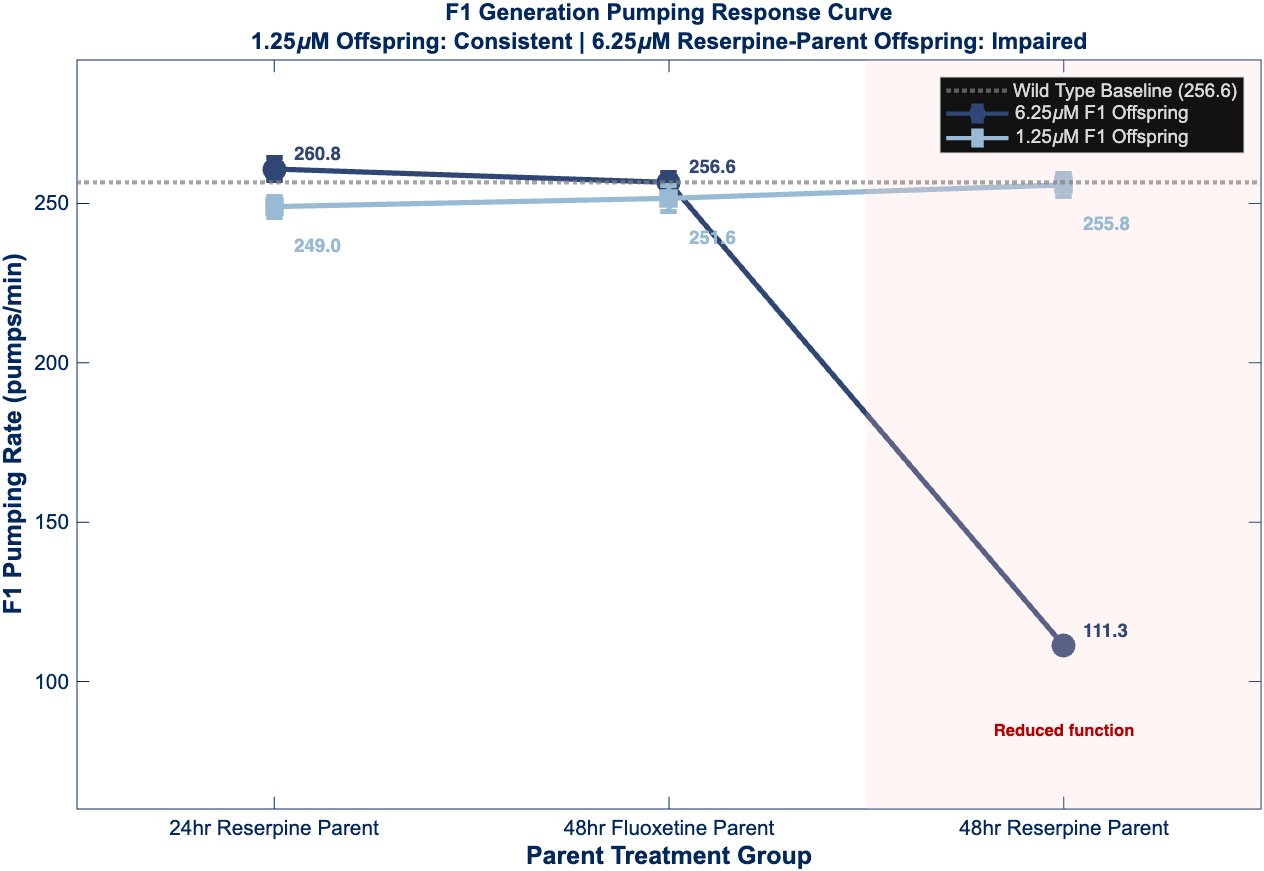 Figure 4 – F1 Generation Transgenerational Effects
Figure 4 – F1 Generation Transgenerational Effects
Response curves and bar graphs demonstrating remarkable transgenerational recovery patterns. The 1.25 µM offspring maintain wild-type behavior across all parental treatment conditions (no significant differences, ns). The 6.25 µM offspring show complete recovery when parents received short-term reserpine or fluoxetine rescue, but exhibit significant impairment (***p < 0.001) when parents were maintained on continuous reserpine.
Dose-Dependent Parental Effects: Statistical analysis confirmed highly significant behavioral impairments in the parental generation across both reserpine dosages. The 6.25 µM group showed severe deficits (t = -19.64 for thrashing, t = -21.26 for pumping, both p < 0.001), while the 1.25 µM group exhibited moderate impairments (t = -12.86 for thrashing, t = -18.31 for pumping, both p < 0.001). This dose-response relationship demonstrates that C. elegans provides a quantitative system for modeling serotonergic dysfunction severity.
Fluoxetine Rescue Efficacy: Fluoxetine treatment produced significant behavioral rescue, though with differential effectiveness. Locomotor rescue was robust across both doses (99.3% and 81.3% baseline recovery), with highly significant improvement over reserpine-only controls (p < 0.001 for both groups). However, pharyngeal pumping showed limited rescue in the high-dose group (1.5% improvement) versus effective rescue in the low-dose group (93.0% recovery), suggesting behavior-specific and dose-dependent response thresholds for SSRI intervention.
Transgenerational Recovery Patterns: The most striking finding was near-complete behavioral normalization in F1 offspring despite persistent parental dysfunction. In the 1.25 µM group, all F1 comparisons showed no significant difference from wild-type (p-values ranging from 0.24 to 1.00), indicating complete developmental compensation. The 6.25 µM group showed mixed results: F1 from 24hr reserpine or fluoxetine-rescued parents achieved wild-type levels (p > 0.05), but offspring from continuously reserpine-treated parents exhibited significant impairment (thrashing: t = -12.07, p < 0.001; pumping: t = -80.11, p < 0.001).
Behavioral Domain Differences: Comparison between thrashing and pumping assays revealed that feeding behavior may be more sensitive to both severe pharmacological disruption and transgenerational effects. The extremely large t-statistic for F1 pumping from 48hr reserpine parents (t = -80.11) was the most robust finding in the entire dataset, suggesting that motivational behaviors controlled by pharyngeal circuits may be particularly vulnerable to inherited effects of severe parental serotonergic dysfunction.
Key Findings: This study demonstrates three principal conclusions: (1) reserpine produces dose-dependent behavioral impairments modeling aspects of depressive symptomatology, with progressive deterioration under chronic exposure; (2) fluoxetine effectively rescues locomotor deficits but shows limited efficacy for feeding behavior in severely affected organisms, suggesting distinct neural mechanisms and the importance of early intervention; and (3) first-generation offspring exhibit near-complete behavioral normalization despite persistent parental dysfunction, with dose-dependent recovery patterns indicating threshold effects in transgenerational programming.
Implications for Hereditary Suicide Risk: These findings challenge deterministic views of hereditary mental health vulnerability. The 55% heritability of suicide risk identified in twin studies may reflect transmission of reversible molecular states rather than immutable genetic programming. The robust transgenerational recovery observed, particularly following moderate parental serotonergic disruption, suggests that developmental compensation mechanisms can effectively buffer offspring against parental neurochemical dysfunction.
Mechanistic Insights: Several mechanisms may contribute to transgenerational recovery, including epigenetic reprogramming during embryogenesis that erases maladaptive chromatin modifications, the maternal-to-zygotic transition that replaces abnormal maternal gene products with zygotic transcripts, and utilization of parallel serotonin biosynthesis pathways less affected by parental reserpine exposure.
Limitations: C. elegans behavioral readouts serve as quantitative proxies rather than direct models of human suicidal ideation. The brief generation time may not capture extended developmental windows during which transgenerational effects operate in humans. This study examined only pharmacological manipulation without directly assessing molecular markers such as serotonin content or epigenetic modifications.
Clinical Relevance: The complete F1 recovery in the low-dose versus partial recovery in the high-dose group implies that early intervention to prevent progression to severe depressive episodes may more effectively protect offspring than treatment after severe symptoms develop. Most importantly, the observation that offspring behavior normalizes despite persistent parental dysfunction provides evidence that hereditary risk is not immutable, supporting preconception and perinatal mental health services as viable primary prevention strategies.
Conclusion: While parental serotonergic dysfunction produces significant behavioral deficits in C. elegans, these effects show limited transgenerational persistence. The robust developmental compensation observed in offspring, particularly following moderate parental disruption, suggests that hereditary risk for mood disorders may be more amenable to modification than traditional genetic models imply.
Cowen, P. J., & Browning, M. (2015). What has serotonin to do with depression? World Psychiatry, 14(2), 158–160. https://doi.org/10.1002/wps.20229
DiBlasi, E. (2021). Serotonin and the improved efficacy of the antidepressant drugs sertraline and mirtazapine in newly diagnosed patients with major depressive disorders. Human Psychopharmacology: Clinical and Experimental. https://doi.org/10.1002/hup.2833
Massachusetts Institute of Technology. (2015). C. elegans synchronization via bleaching (7.15 Experimental Molecular Genetics, Spring 2015). MIT OpenCourseWare. https://ocw.mit.edu
Niacaris, T. (2003). Serotonin regulates repolarization of the C. elegans pharyngeal muscle. Journal of Experimental Biology, 206(2), 223–231. https://doi.org/10.1242/jeb.00101
Pandey, G. N. (2013). Biological basis of suicide and suicidal behavior. Bipolar Disorders, 15(5), 524–541. https://doi.org/10.1111/bdi.12089
Ranganathan, R., Sawin, E. R., Trent, C., & Horvitz, H. R. (2001). Mutations in the Caenorhabditis elegans Serotonin Reuptake Transporter MOD-5 Reveal Serotonin-Dependent and -Independent Activities of Fluoxetine. The Journal of Neuroscience, 21(16), 5871–5884. https://doi.org/10.1523/jneurosci.21-16-05871.2001
Sun, S., Liu, Q., Wang, Z., Huang, Y., Sublette, M. E., Dwork, A. J., Rosoklija, G., Ge, Y., Galfalvy, H., Mann, J. J., & Haghighi, F. (2024). Brain and blood transcriptome profiles delineate common genetic pathways across suicidal ideation and suicide. Molecular Psychiatry, 1–10. https://doi.org/10.1038/s41380-024-02420-z
Voracek, M., & Loibl, L. M. (2007). Genetics of suicide: a systematic review of twin studies. Wiener Klinische Wochenschrift, 119(15-16), 463–475. https://doi.org/10.1007/s00508-007-0823-2
Yu, J., Vogt, M. C., Fox, B. L., Wrobel, C. J. J., Diana Fajardo Palomino, Curtis, B. J., Zhang, B., Le, H. H., Arnaud Tauffenberger, Hobert, O., & Schroeder, F. C. (2022). Parallel pathways for serotonin biosynthesis and metabolism in C. elegans. 19(2), 141–150. https://doi.org/10.1038/s41589-022-01148-7
Research Poster: A comprehensive visual summary of the experimental design, methodology, results, and conclusions. The poster presents the transgenerational effects of parental serotonergic modulation in C. elegans, highlighting dose-dependent behavioral impairments, differential fluoxetine rescue efficacy, and remarkable offspring recovery patterns. Key figures include time-course graphs, statistical comparisons, and transgenerational response curves demonstrating threshold effects in developmental compensation mechanisms. The poster emphasizes clinical implications for understanding hereditary suicide risk and the potential for preventive interventions targeting parental mental health.