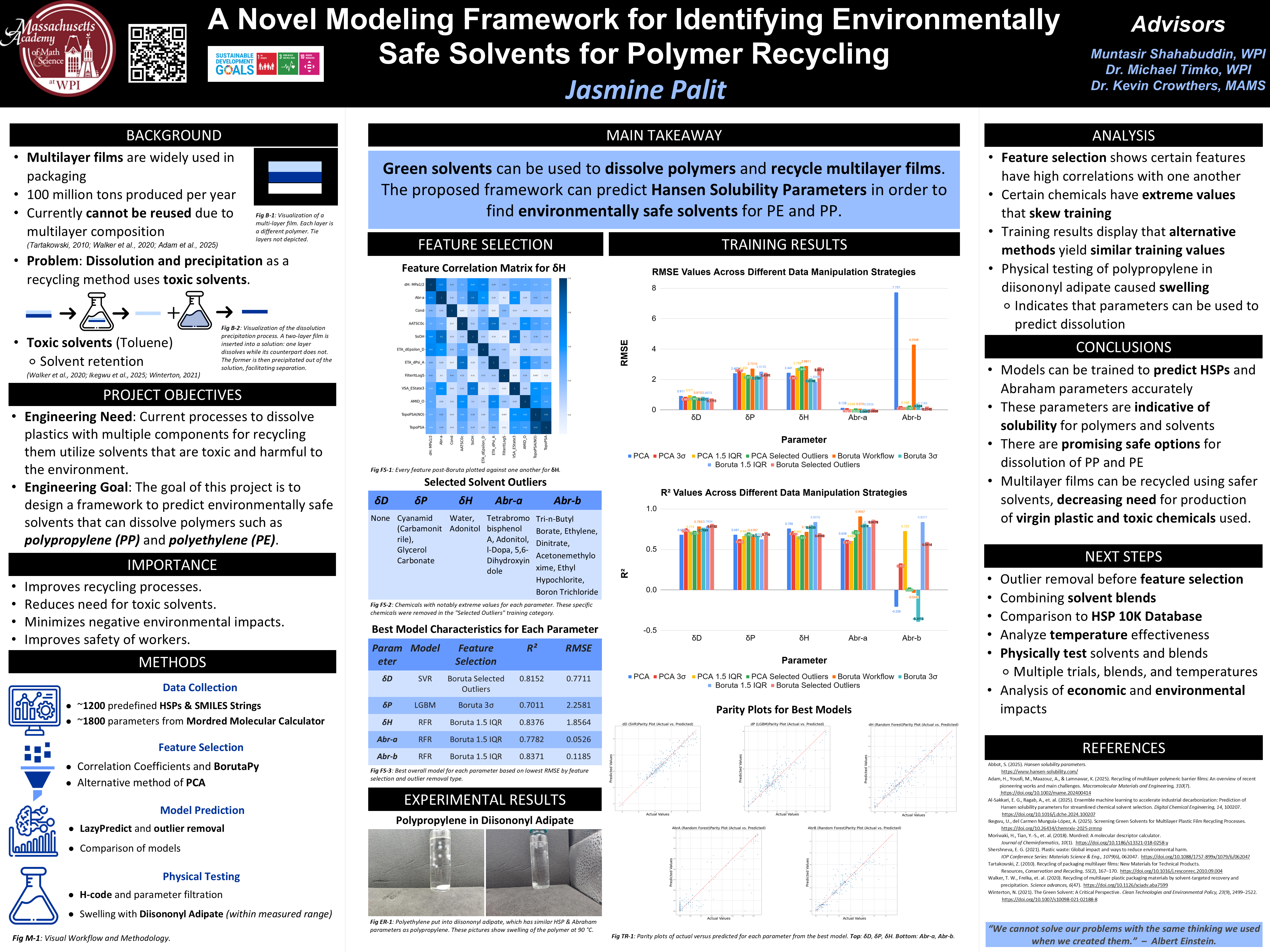
In STEM, we learn about how to conduct scientific research and present our findings in a clear and organized manner. We learn to understand published journal articles, write grant proposals, and present our research to a variety of audiences. Currently, we are working on our independent research project, an overview of which is shown below.
Main Takeaway
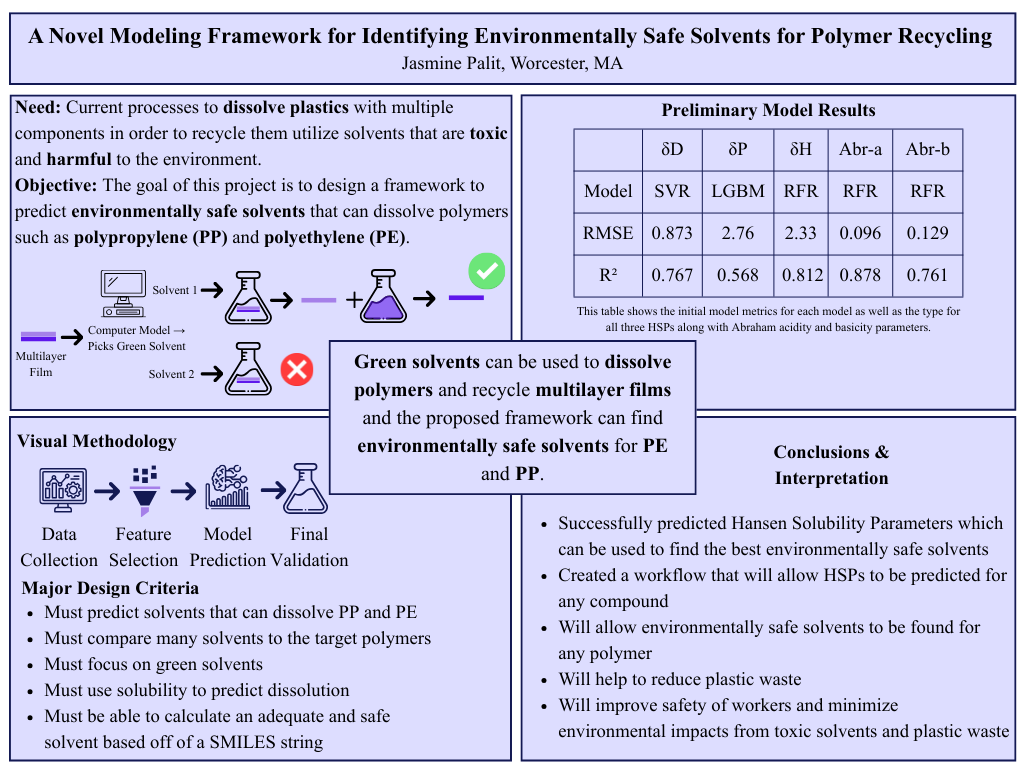
Green solvents can be used to dissolve polymers and recycle multilayer films. The proposed framework can predict Hansen Solubility Parameters in order to find environmentally safe solvents for PE and PP.
Abstract
Plastic damages the environment and human health through its production and end-of-life effects. To combat this, recycling is used to repurpose old plastics into new products. However, some common plastics, like multilayer films, have multiple types of polymers in a distinct layered structure. Thus, to recycle them, the layers must first be separated before they can be reused. One method to accomplish this goal is through sequential dissolution and precipitation.
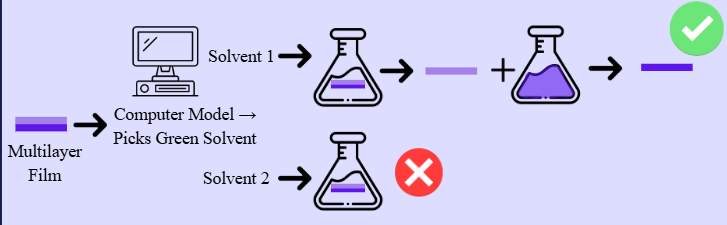
During dissolution and precipitation, one polymer is dissolved while the others remain solid, separating them. While this technique is effective, the solvents used are often harmful to the environment and workers, such as using Toluene to dissolve polyethylene (PE) and polypropylene (PP). Thus, more environmentally safe solvents are needed in recycling processes.
Given that PE and PP presently are quite common in multilayer films, a safer solvent is necessary. To accomplish this, Hansen Solubility Parameters are utilized to decide which solvents PE and PP are miscible in. However, these parameters are not defined for all green solvents. Machine learning models are trained to predict each HSP, with R-squared values ranging between 0.7 and 0.9. The parameters are compared to the target polymer to determine which green solvent is most effective, potentially validated through physical testing.
The implications of this project are creating a healthier planet, reducing human health risks from working with toxic solvents, and decreasing the quantity of discarded and produced plastic. The process can eventually be applied to all polymers that require safe solvents, creating safer recycling processes.
For more information, see the project proposal and documents here.
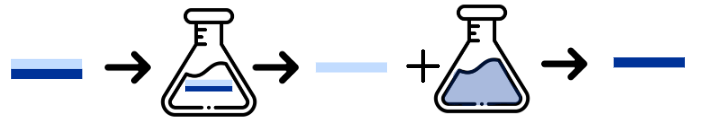
Visualization of the dissolution precipitation process. A two-layer film is inserted into a solution: one layer dissolves while its counterpart does not. The former is then precipitated out of the solution, facilitating separation.
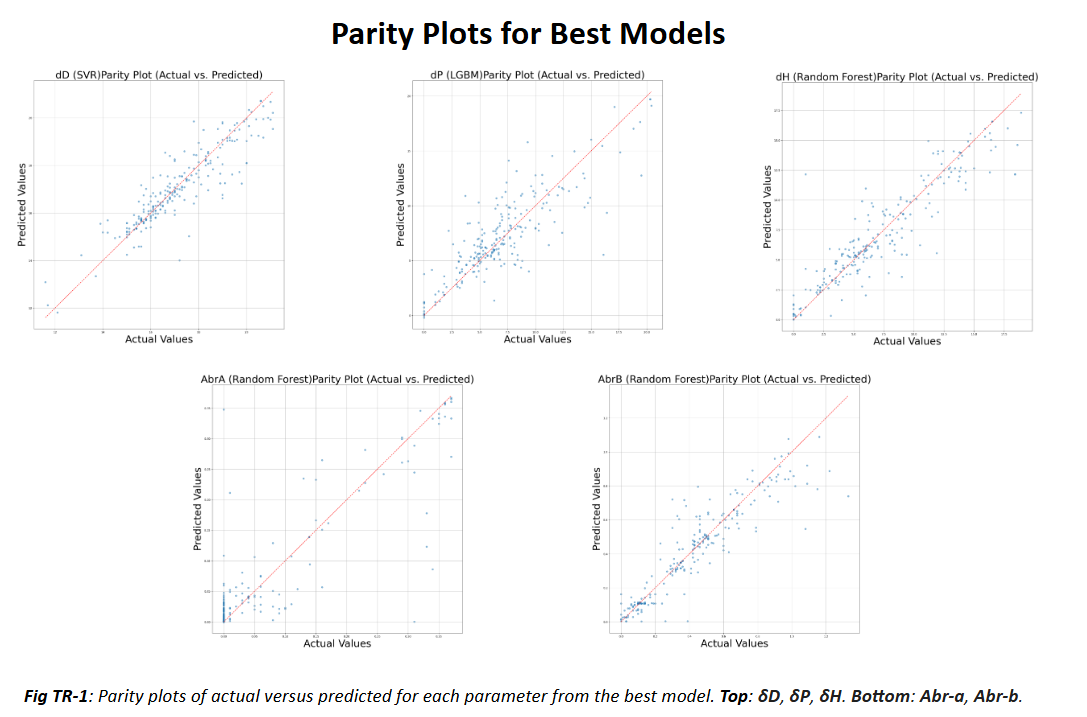
To determine which solvents are will successfully dissolve the target polymers, Hansen Solubility Parameters (HSP) are used. HSP are a set of three parameters that describe the solubility of a material in a solvent. They are based on the idea that "like dissolves like," meaning that a solvent will dissolve a solute if their HSP values are similar. The three parameters are the dispersion parameter (δD), the polar parameter (δP), and the hydrogen bonding parameter (δH). By comparing the HSP values of the target polymer to those of potential solvents, we can predict which solvents will be effective for dissolution. However, HSP values are not available for all solvents, especially green solvents. To address this issue, machine learning models are trained to predict the HSP values for a range of solvents, including green solvents. The predicted HSP values are then compared to the HSP values of the target polymers to identify which solvents are most likely to be effective for dissolution. The predictions can then be validated through physical testing to confirm the effectiveness of the identified solvents.
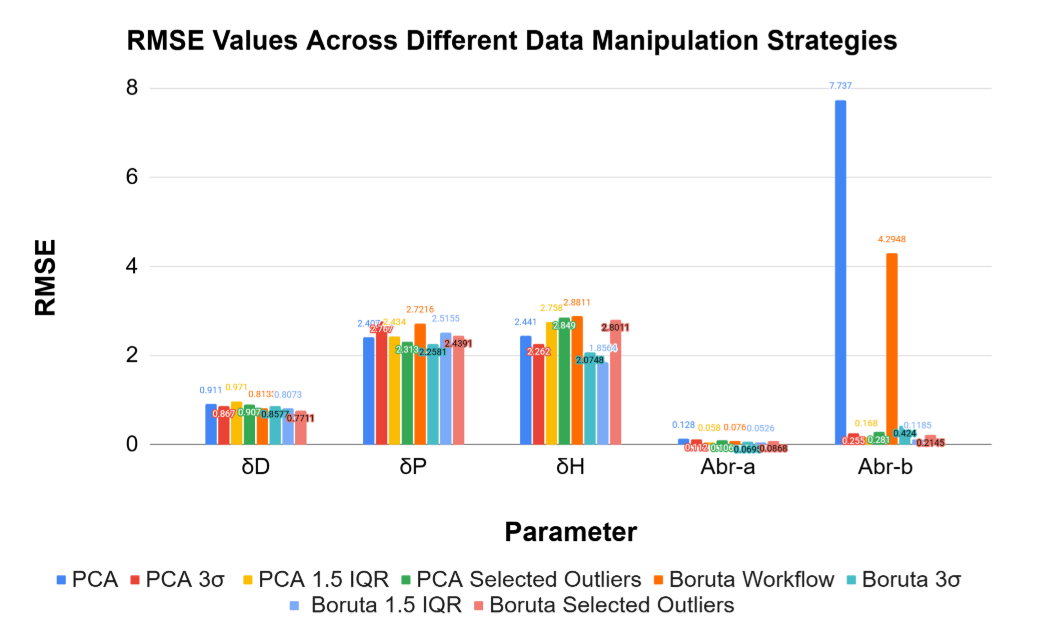
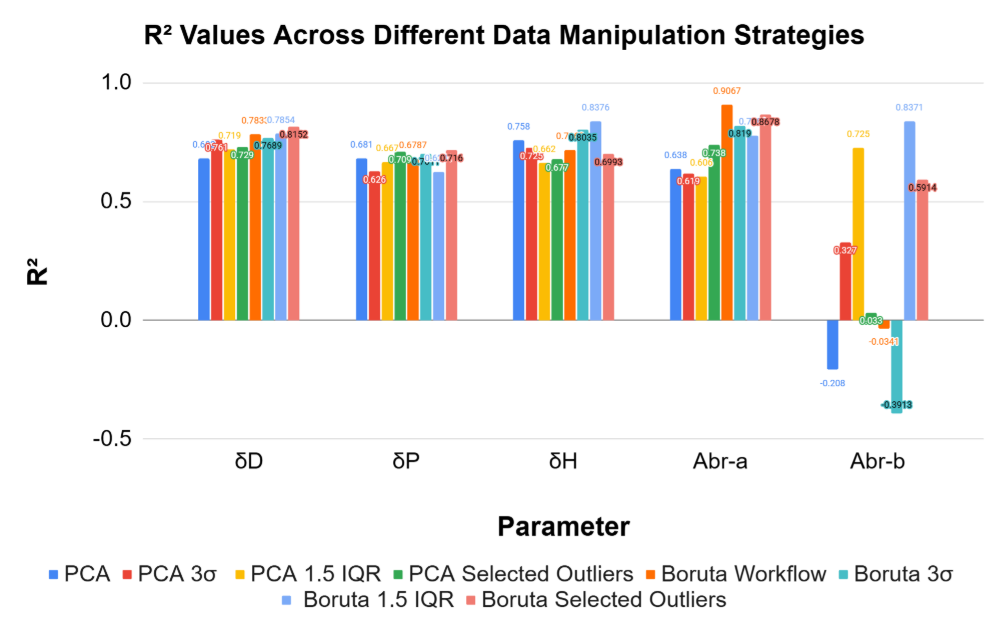
To create the best possible model for prediction, two different feature selection methods and 3 different outlier removal methods were used. The Boruta models used correlation sorting and BorutaPy for feature selection, choosing around 10-20 features for each target variable. Alternatively, principal component analysis (PCA) was used to reduce the dimensionality of the data, with the number of components chosen to explain 95% of the variance in the data. For outlier removal, three different methods were used: interquartile range (IQR), standard deviation, and selection. The IQR method identifies outliers as data points that fall outside of 1.5 times the interquartile range above the third quartile or below the first quartile. The Z-score method identifies outliers as data points that have a Z-score greater than 3 or less than -3. The selected outliers were obvious outliers from looking at the dataset. By using these different feature selection and outlier removal methods, we can create multiple models and compare their performance to identify which combination yields the best results for predicting HSP values.
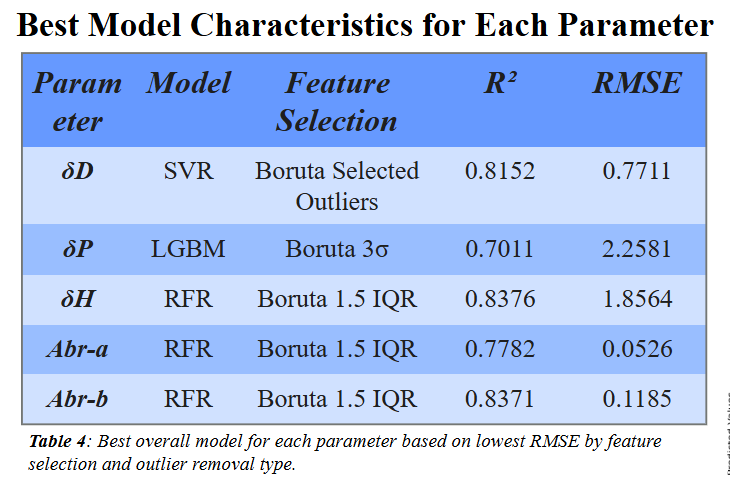
Feature selection shows certain features have high correlations with one another
Certain chemicals have extreme values that skew training
Training results display that alternative methods yield similar training values
Physical testing of polypropylene in diisononyl adipate caused swelling
Indicates that parameters can be used to predict dissolution
Models can be trained to predict HSPs and Abraham parameters accurately
These parameters are indicative of solubility for polymers and solvents
There are promising safe options for dissolution of PP and PE
Multilayer films can be recycled using safer solvents, decreasing need for production of virgin plastic and toxic chemicals used.
References
Abbott, S. (2025). Hansen solubility parameters. https://www.hansen-solubility.com/
Adam, H., Yousfi, M., Maazouz, A., & Lamnawar, K. (2025). Recycling of multilayer polymeric barrier films: An overview of recent pioneering works and main challenges. Macromolecular Materials and Engineering, 310(7). https://doi.org/10.1002/mame.202400414
Al-Sakkari, E. G., Ragab, A., et al. (2025). Ensemble machine learning to accelerate industrial decarbonization: Prediction of Hansen solubility parameters for streamlined chemical solvent selection. Digital Chemical Engineering, 14, 100207. https://doi.org/10.1016/j.dche.2024.100207
Ikegwu, U., & del Carmen Munguía-López, A. (2025). Screening green solvents for multilayer plastic film recycling processes. ChemRxiv. https://doi.org/10.26434/chemrxiv-2025-zrmnp
Moriwaki, H., Tian, Y.-S., Kawashita, N., & Takagi, T. (2018). Mordred: A molecular descriptor calculator. Journal of Cheminformatics, 10(1). https://doi.org/10.1186/s13321-018-0258-y
Shershneva, E. G. (2021). Plastic waste: Global impact and ways to reduce environmental harm. IOP Conference Series: Materials Science and Engineering, 1079(6), 062047. https://doi.org/10.1088/1757-899X/1079/6/062047
Tartakowski, Z. (2010). Recycling of packaging multilayer films: New materials for technical products. Resources, Conservation and Recycling, 55(2), 167–170. https://doi.org/10.1016/j.resconrec.2010.09.004
Walker, T. W., Frelka, N., et al. (2020). Recycling of multilayer plastic packaging materials by solvent-targeted recovery and precipitation. Science Advances, 6(47). https://doi.org/10.1126/sciadv.aba7599
Winterton, N. (2021). The green solvent: A critical perspective. Clean Technologies and Environmental Policy, 23(9), 2499–2522. https://doi.org/10.1007/s10098-021-02188-8