Background Description
Alzheimer's Disease (AD) is a devastating neurodegenerative disorder affecting over 6 million Americans and ranking as the seventh leading cause of death in the United States. The disease progressively destroys memory, thinking skills, and behavioral regulation through the accumulation of toxic proteins such as amyloid-beta (Aβ) and tau in the brain. While AD predominantly affects elderly individuals—likely due to age-related inflammation, blood vessel damage, and brain atrophy—emerging research reveals a complex relationship between metabolic dysfunction and neurodegeneration. This connection has led many in the medical community to describe Alzheimer's as "Type 3 Diabetes," reflecting the critical role of insulin resistance in the disease's progression. When insulin resistance develops, insulin degrading enzymes (IDEs) become overwhelmed breaking down excess insulin rather than clearing Aβ peptides, leading to toxic protein accumulation and subsequent neurodegeneration.
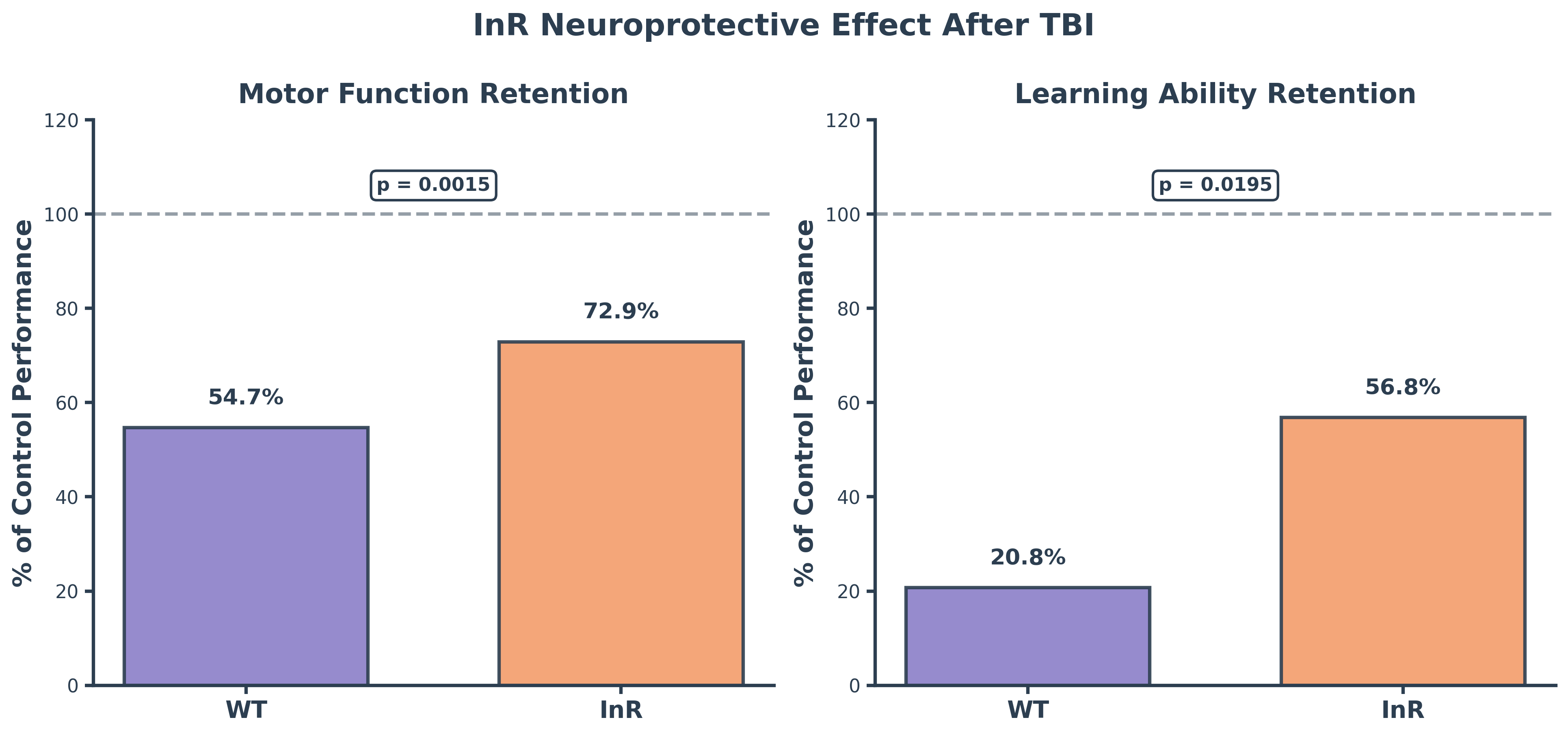
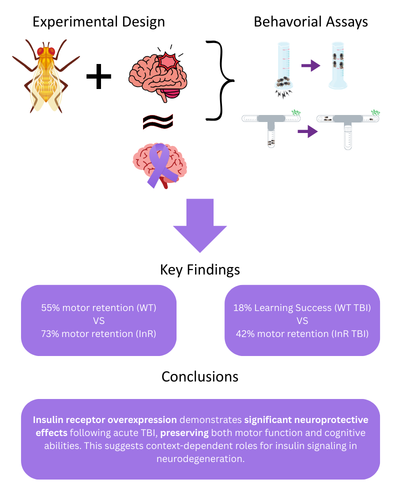
The role of insulin signaling in the brain presents a fascinating paradox. While insulin resistance clearly contributes to Alzheimer's pathology, research in Drosophila melanogaster reveals that genetically reducing insulin signaling pathway (ISP) activity can actually protect against Aβ toxicity. Studies have shown that knocking down chico—a gene encoding the fruit fly equivalent of Insulin Receptor Substrate-1 that bridges the insulin receptor to downstream signaling components—significantly improves locomotor ability and reduces brain vacuolization in flies expressing toxic Aβ protein. Similar neuroprotective effects occur when other key ISP components like dInR or PI3K are knocked out. This protection appears to work by activating stress-response pathways and increasing autophagy, creating a cellular environment more resistant to protein toxicity. Conversely, chronically elevating insulin signaling through InR overexpression reduces autophagy, promotes excessive cellular growth, lowers stress resistance, and in some cases worsens neurodegenerative symptoms, suggesting that the optimal level of insulin signaling for brain health may fall within a critical range.
Multiple pathological triggers can initiate Alzheimer's-like neurodegeneration, making it essential to test potential interventions across different disease models. High-sugar diets cause metabolic dysregulation leading to Type 2 diabetes, which represents one of the biggest risk factors for AD alongside aging itself. Traumatic brain injury (TBI) presents another powerful trigger, as individuals sustaining moderate to severe TBI show significantly elevated risk of dementia later in life. Following injury, the brain exhibits chronic inflammation, tau hyperphosphorylation, oxidative stress, and impaired clearance of toxic proteins—mechanisms that closely mirror the pathological cascade of AD. These overlapping mechanisms make TBI particularly valuable for creating Alzheimer's-relevant models, as it can initiate or exacerbate AD-like neurodegeneration in genetically or metabolically vulnerable individuals through ethically accessible experimental procedures.
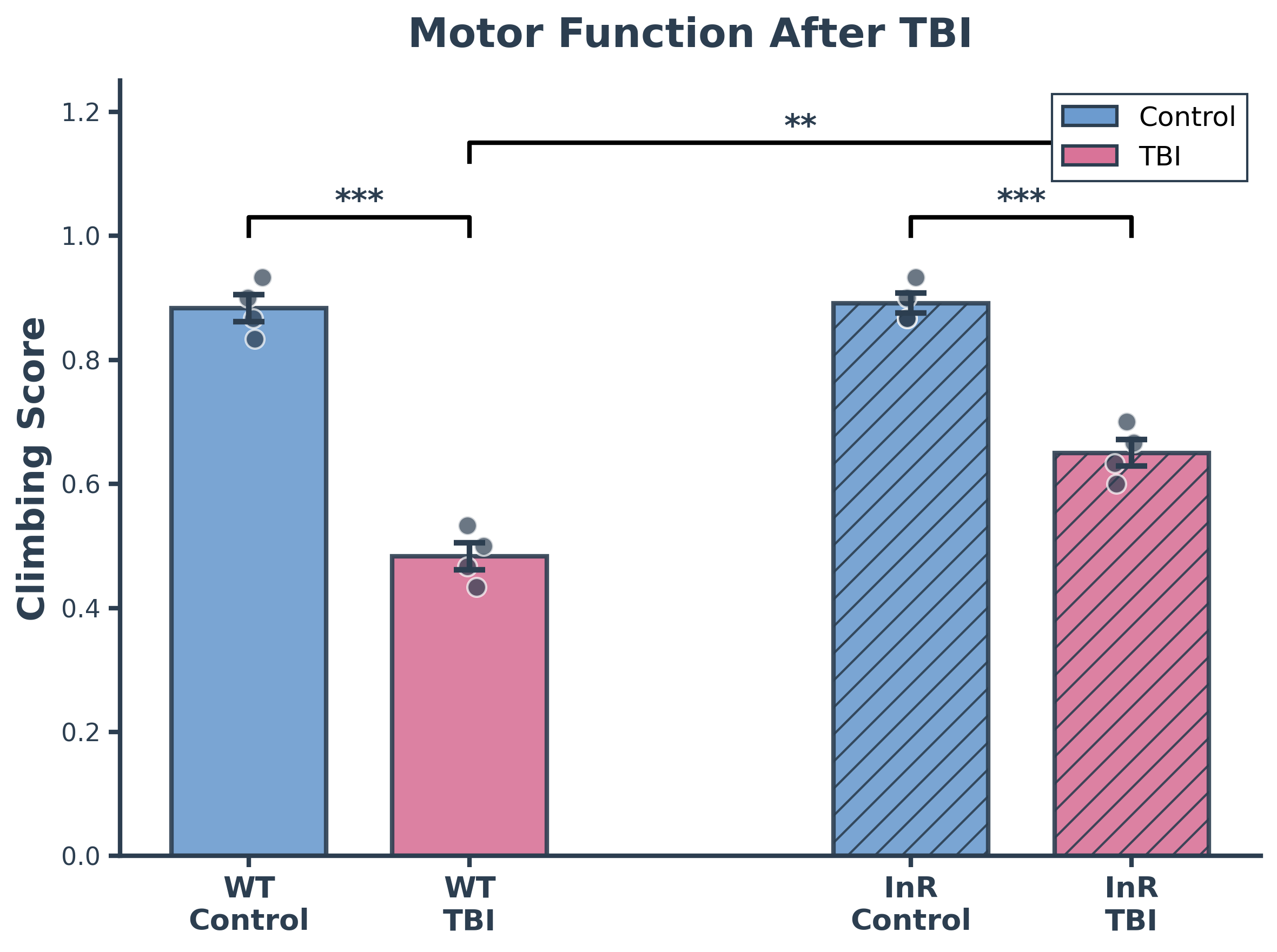
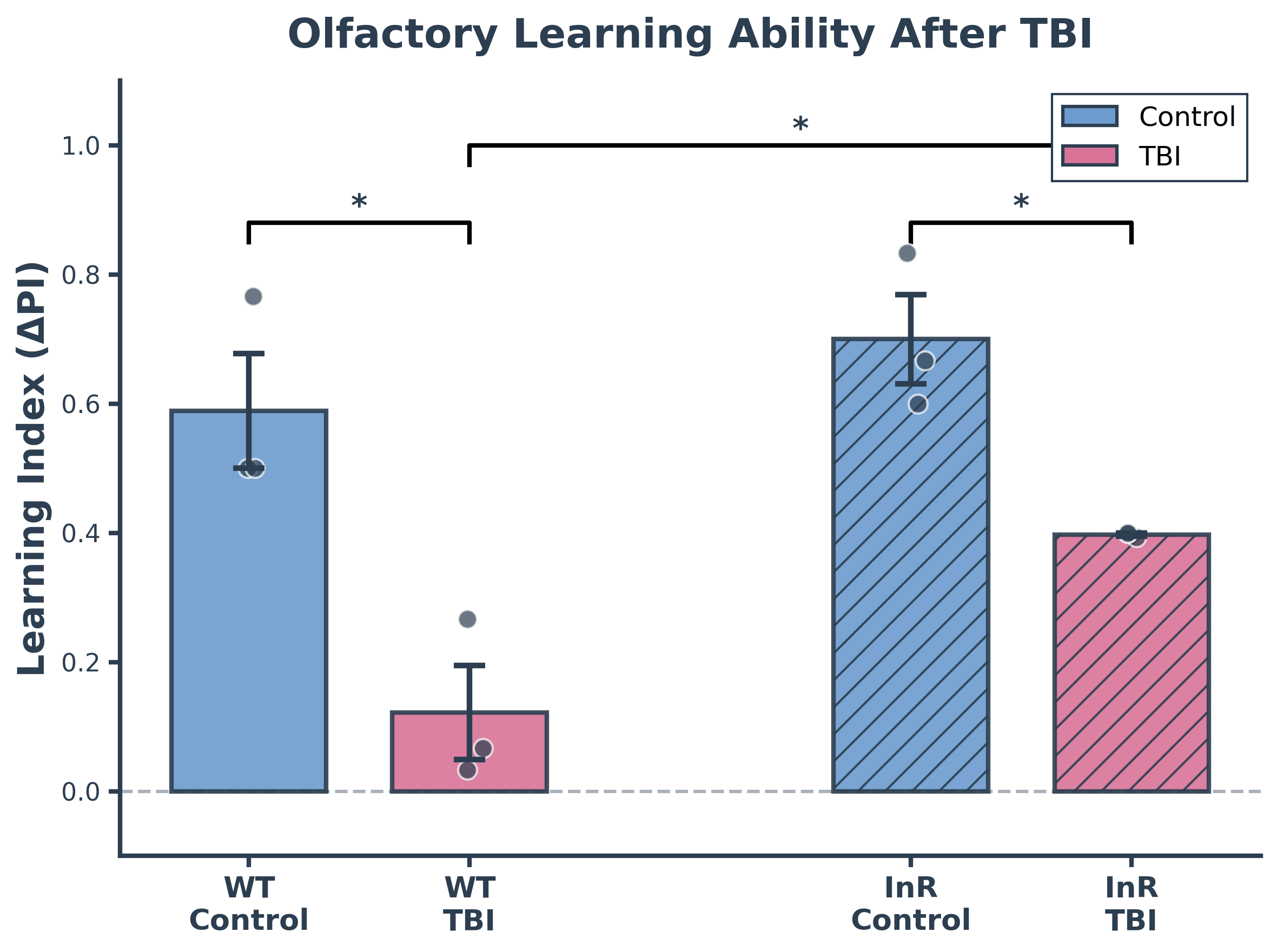
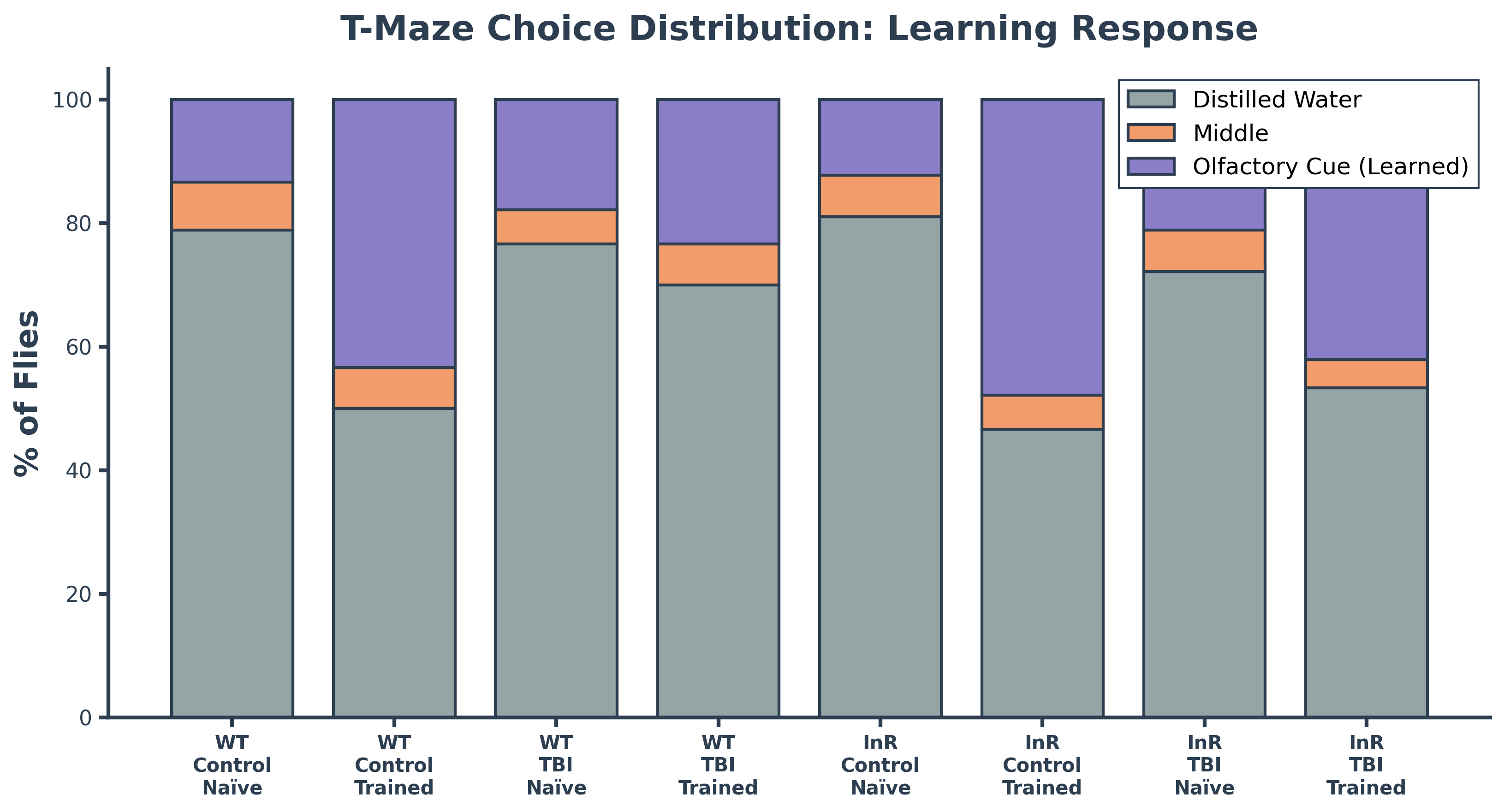
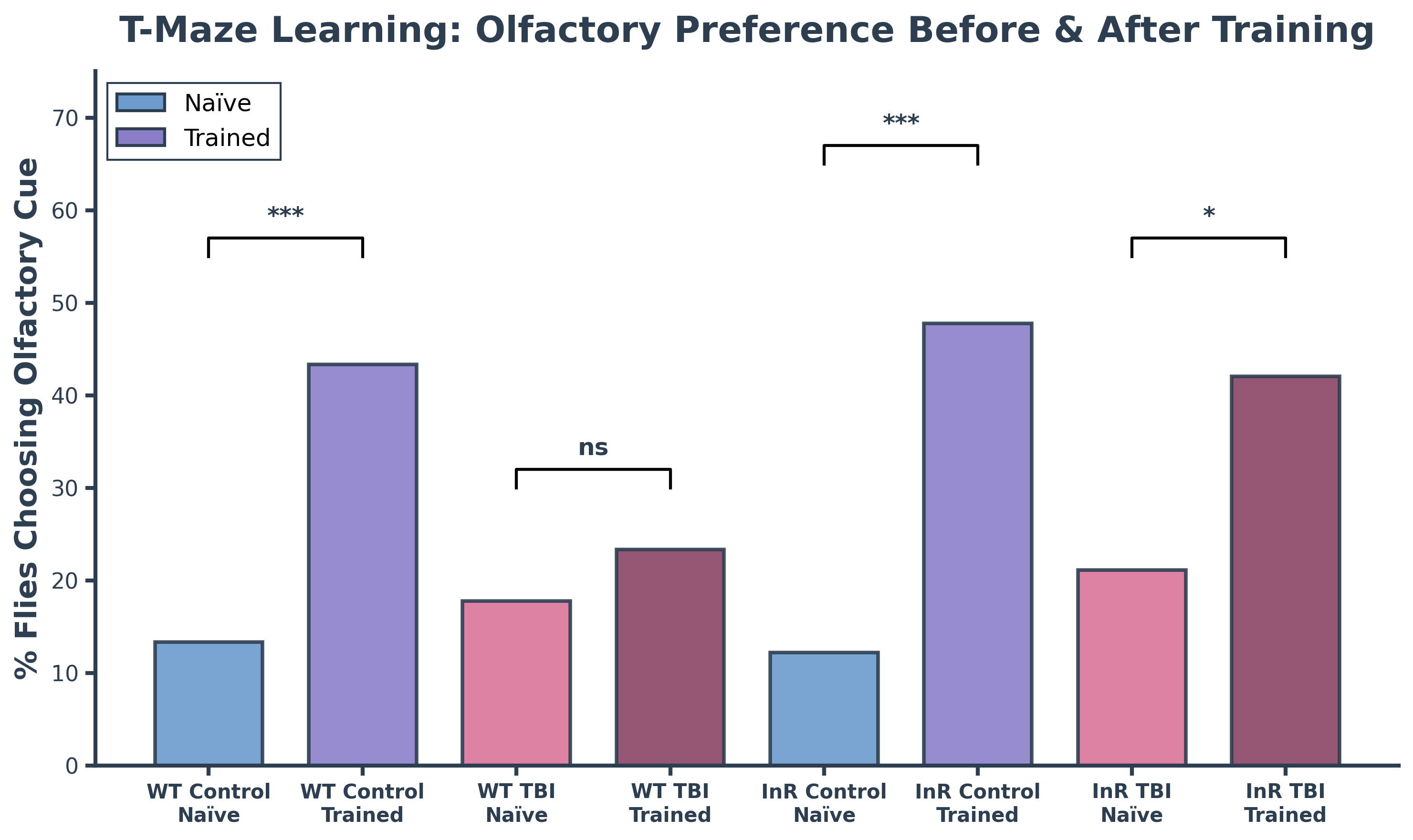
Drosophila melanogaster serves as an ideal organism for investigating these complex relationships between insulin signaling and neurodegeneration. Fruit flies offer significant advantages over alternative model organisms: they lack the ethical concerns associated with vertebrate research, can be maintained cheaply in large quantities, and possess highly conserved signaling pathways and cellular processes that directly translate to human biology. Neurodegeneration in flies manifests through measurable declines in locomotion, memory, and behavioral regulation—all mediated by neural circuits remarkably similar to those in humans. Locomotor deficits assessed through climbing assays reflect motor neuron dysfunction, neuromuscular weakness, and mitochondrial decline. Memory and learning capacity can be quantified using olfactory T-maze assays, where flies choose between arms containing isoamyl acetate (an initially aversive odorant) versus distilled water; naïve flies avoid the odorant while conditioned flies learn it's harmless, with the shift in distribution revealing learning capacity. Aggressive behavior, measured through controlled male-male interactions, provides insight into higher-order behavioral dysfunction caused by neural impairment. Despite extensive research demonstrating that ISP modulation affects Aβ toxicity, the differential effects of reducing versus enhancing insulin signaling across multiple neurodegenerative markers and various AD-relevant stressors remain incompletely understood, creating an opportunity to identify potential therapeutic strategies for combating neurodegeneration.
.png)